Lesson 6
Acute Renal Failure
Acute renal failure occurs in 5
percent of hospitalized patients. Etiologically, this common condition can be
categorized as prerenal, intrinsic or postrenal.

Most patients have prerenal acute
renal failure or acute tubular necrosis (a type of intrinsic acute renal
failure that is usually caused by ischemia or toxins). Using a systematic approach, physicians can determine
the cause of acute renal failure in most patients. This approach includes a
thorough history and physical examination, blood tests, urine studies and a
renal ultrasound examination. In certain situations, such as when a patient has
glomerular disease, microvascular disease or obstructive disease, rapid
diagnosis and treatment are necessary to prevent permanent renal damage. By
maintaining euvolemia, recognizing patients who are at increased risk and
minimizing exposure to nephrotoxins, physicians can decrease the incidence of
acute renal failure. Once acute renal failure develops, supportive therapy is
critical to maintain fluid and electrolyte balances, minimize nitrogenous waste
production and sustain nutrition. Death is most often caused by infection or
cardiorespiratory complications
Oliguria
literally means "reduced" urine volume— less than that necessary to
remove endogenous solute loads that are the end products of metabolism. As long
as the patient's kidney can usually concentrate urine in a normal fashion to a
specific gravity of 1.035, oliguria (for that person) is present at urine
volumes under 400 mL/d, or approximately 6 mL/kg body weight. On the other
hand, if the kidney concentration is impaired and the patient can achieve a
specific gravity of only 1.010, oliguria is present at urine volumes under
1000-1500 mL/d.
Acute
renal failure is a condition in which the glomerular filtration rate is
abruptly reduced, causing a sudden retention of endogenous and exogenous
metabolites (urea, potassium, phosphate, sulfate, creatinine, administered
drugs) that are normally cleared by the kidneys. The urine volume is usually
low (under 400 mL/d). However, if renal concentrating mechanisms are impaired
during acute renal failure, the daily urine volume may be normal or even high (high-output
or nonoliguric renal failure). Rarely, there is no
urine output at all (anuria) in acute renal failure. 
Prompt differentiation of the cause is important
in determining appropriate therapy. Prerenal renal failure is reversible if
treated promptly, but a delay in therapy may allow it to progress to a fixed,
nonspecific form of intrinsic renal failure (eg, acute tubular necrosis). The
other causes of acute renal failure are classified on the basis of their
involvement with vascular lesions, intrarenal disorders, or postrenal
disorders. 

PRERENAL
RENAL FAILURE
The
term prerenal denotes inadequate renal perfusion because of reduced
intravascular volume or lowered effective arterial circulation. The most
common cause of this form of acute renal failure is dehydration due to renal
or extrarenal fluid losses from diarrhea, vomiting, excessive use of
diuretics, and so on.
Less
common causes are septic shock, "third spacing" with extravascular
fluid pooling (eg, pancreatitis), and excessive use of antihypertensive drugs,
which causes relative or absolute depletion of intravascular fluid volume.
Heart failure with reduced cardiac output also can reduce effective renal blood
flow. Careful clinical assessment may identify the primary condition
responsible for prerenal renal failure, but many times several conditions can
coexist. In the hospital setting, these circulatory abnormalities often lead to
more fixed, acute renal failure (acute tubular necrosis), since these patients
often have multiple comorbid conditions.
Acute
reductions in glomerular filtration rate may also be noted in patients with cirrhosis
(hepatorenal failure) or in patients taking cyclosporine, tacrolimus,
nonsteroidal antiinflammatory drugs, or angiotensin-converting enzyme
inhibitors. It is felt that these conditions represent significant intrarenal
hemodynamic functional derangements mediated by prostaglandins and
renin-angiotensin such that the glomerular capillary pressure suddenly falls.
In these clinical circumstances, the urinary findings may mimic prerenal renal
failure, but the patient's clinical assessment does not demonstrate the
extrarenal findings seen in common prerenal conditions, as noted in the
following section. Improvements in glomerular filtration rate are usually noted
after drug discontinuance or, in cases of hepatorenal renal failure, with
management of the liver disease or liver transplantation.
Clinical
Findings
A.
Symptoms and Signs: Except for rare cases with associated
cardiac or "pump" failure, patients usually complain of thirst or of
dizziness in the upright posture (orthostatic dizziness). There may be a
history of overt fluid loss. Weight losses over hours to days quantitatively
reflect the degree of dehydration.
Physical
examination frequently reveals decreased skin turgor, collapsed neck veins, dry
mucous membranes and axillas, and, most important, orthostatic or postural
changes in blood pressure and pulse.

Table 1 Causes of acute
renal failure.
I. Prerenal
renal failure:
1.
Dehydration
2.
Vascular collapse due to sepsis, antihypertensive drug therapy, "third
spacing"
3.
Reduced cardiac output
II. Functional-hemodynamic:
1.
Angiotensin-converting enzyme inhibitor drugs
2.
Nonsteroidal anti-inflammatory drugs
3.
Cyclosporine; tacrolimus
4.
Hepatorenal syndrome
III. Vascular:
1.
Atheroembolism
2.
Dissecting arterial aneurysms
3.
Malignant hypertension
IV. Parenchymal
(intrarenal):
1.
Specific:
a.
Glomerulonephritis
b.
Interstitial nephritis
c.
Toxin, dye-induced
d.
Hemolytic uremic syndrome
2.
Nonspecific:
a.
Acute tubular necrosis
b.
Acute cortical necrosis
V.
Postrenal:
1.
Calculus in patients with solitary kidney
2.
Bilateral ureteral obstruction
3.
Outlet obstruction
4.
Leak, post-traumatic
B.
Laboratory Findings:
1. Urine
The urine volume is
usually low. Accurate assessment may require bladder catheteriza-tion followed
by hourly output measurements (which will also rule out lower urinary tract
obstruction; see discussion following). High urine specific gravity (>
1.025) and urine osmolality (> 600 mOsm/kg) also are noted in this form of
acute apparent renal failure. Routine urinalysis usually shows no
abnormalities.
2. Urine
and blood chemistries-The blood urea nitrogen-creatinine ratio, normally 10:1, is usually increased with prerenal renal failure.
Because mannitol, radiocontrast dyes, and diuretics affect the delivery and
tubular handling of urea, sodium, and creatinine, urine and blood chemistry
tests performed after these agents have been given produce misleading results.
3. Central
venous pressure-A low central venous pressure indicates hypovolemia, which
may be due to blood loss or dehydration. If severe cardiac failure is the
principal cause of prerenal renal failure (it is rarely the sole cause),
reduced cardiac output and high central venous pressure are apparent.
4. Fluid
challenge-An increase in urine output in response to a carefully
administered fluid challenge is both diagnostic and therapeutic in cases of
prerenal renal failure. Rapid intravenous administration of 300-500 mL of
physiologic saline or 125 mL of 20% mannitol (25 g/125 mL) is the usual initial
treatment. Urine output is measured over the subsequent 1-3 h. A urine volume
increase of more than 50 mL/h is considered a favorable response that warrants
continued intravenous infusion with physiologic solutions to restore plasma
volume and correct dehydration. If the urine volume does not increase, the
physician should carefully review the results of blood and urine chemistry
tests, reassess the patient's fluid status, and repeat the physical examination
to determine whether an additional fluid challenge (with or without
furosemide) might be worthwhile.
Treatment
In
states of dehydration, measured and estimated fluid losses must be rapidly
corrected to treat oliguria of prerenal origin. Inadequate fluid management may
cause further renal hemodynamic deterioration and eventual renal tubular
ischemia (with fixed acute tubular necrosis; see discussion following). If
oliguria persists in a well-hydrated patient, vasopressor drugs are indicated
in an effort to correct the hypotension associated with sepsis or cardiogenic
shock. Pressor agents that restore systemic blood pressure while maintaining
renal blood flow and renal function are most useful. Dopamine, 1-5 µg/kg/min,
gives "renal-dose" levels providing an increase in renal blood flow
without systemic pressor responses. Higher doses of 5-20 µg/kg may be
necessary if systemic hypotension persists after volume correction.
Discontinuance of antihypertensive medications or diuretics can, by itself,
cure the apparent acute renal failure resulting from prerenal conditions.

VASCULAR
RENAL FAILURE
Common
causes of acute renal failure due to vascular disease include atheroembolic
disease, dissecting arterial aneurysms, and malignant hypertension.
Atheroembolic disease is rare before age 60 and in patients who have not
undergone vascular procedures or angiographic studies. Dissecting arterial
aneurysms and malignant hypertension are usually clinically evident. Acute
renal venous thrombosis, unless it affects both kidneys, has no deleterious
effect on renal clearance function.
Rapid
assessment of the arterial blood supply to the kidney requires arteriography or
other renal blood flow studies (eg, magnetic resonance imaging or Doppler
ultrasound). The cause of malignant hypertension may be identified on physical
examination (eg, scleroderma). Primary management of the vascular process is
necessary to affect the course of these forms of acute renal failure.
INTRARENAL
Diseases
in this category can be divided into specific and nonspecific parenchymal
processes.
1.
SPECIFIC INTRARENAL DISEASE STATES
The
most common causes of intrarenal acute renal failure are acute or rapidly
progressive glomerulonephritis, acute interstitial nephritis, toxic
nephropathies, and hemolytic uremic syndrome.
Clinical
Findings
A.
Symptoms and Signs: Usually
the history shows some salient data such as sore throat or upper respiratory
infection, diarrheal illness (occasionally as epidemic), use of antibiotics, or
intravenous use of drags (often illicit types).
 Oedema
of the lung
Oedema
of the lung

Bilateral
back pain, at times severe, is occasionally noted. Gross hematuria may be
present. It is unusual for pyelonephritis to present as acute renal failure
unless there is (1) associated sepsis or dehydration, (2) obstruction, or (3)
involvement of a solitary kidney. Systemic diseases in which acute renal
failure occurs include Henoch-Schonlein purpura, systemic lupus erythematosus,
and scleroderma. Human immunodeficiency virus (HIV) infection may present with
acute renal failure from HIV nephropathy. The prognosis is quite poor with this
condition.
 Henoch-Schonlein purpura
Henoch-Schonlein purpura
B. Laboratory
Findings:
1. Urine-Urinalysis discloses variably
active sediments: many red and white cells and multiple types of cellular and
granular casts (telescopic urine). Phase contrast microscopy usually reveals
dysmorphic red cells in the urine. In allergic interstitial nephritis,
eosinophils may be noted. The urine sodium concentration may range from 10 to 40 mEq/L.
2. Blood
test- Components of serum complement are often diminished during
deposition of immune complexes. In a few conditions, circulating immune
complexes can be identified. Other tests may disclose systemic diseases such as
lupus erythematosus. Thrombocytopenia and altered red cell morphologic
structure are noted in peripheral blood smears in the hemolytic uremic syndrome
(HUS).
3.
Renal biopsy- Biopsy examination shows characteristic
changes of glomerulonephritis, acute interstitial nephritis, or glomerular
capillary thrombi (in HUS). There may be extensive crescents involving
Bowman's space.
C.
X-Ray Findings: Poor visualization on intravenous urography or
radionuclide renal scans is characteristic. Routine intravenous urography
should be avoided because of the risk of dyeinduced renal injury.
For this reason, sonography is preferable to
rule out obstruction.

 CT
CT
Treatment 
Therapy
is directed toward eradication of infection, removal of antigen, elimination of
toxic materials and drugs, suppression of autoimmune mechanisms, removal of
autoimmune antibodies, or a reduction in effector-inflammatory responses.
Immunotherapy may involve drugs, anticoagulants, or the temporary use of
plasmapheresis. Initiation of supportive dialysis may be required.
 Dialysis
Dialysis

2.
NONSPECIFIC INTRARENAL STATES
Nonspecific
intrarenal causes of acute renal failure include acute tubular necrosis and
acute cortical necrosis. The latter presents with anuria and associated
intrarenal intravascular coagulation and generally has a poorer prognosis than
the former. These forms of acute renal failure usually occur in hospital
settings. Various morbid conditions leading to septic syndrome-like physiologic
disturbances are often present.
Acute
tubular necrosis was initially described by Liicke during World War II in
patients suffering crush injuries and shock. Degenerative changes of the distal
tubules (lower nephron nephrosis) were believed to be due to ischemia. When
dialysis became available, most of these patients recovered—sometimes
completely—provided intrarenal intravascular coagulation and cortical necrosis
had not occurred.
Elderly
patients are more prone to develop this form of oliguric acute renal failure
following hypotensive episodes. It appears that exposure to some drugs (eg,
prostaglandin inhibitors such as nonsteroidal antiinflammatory agents) may
increase the risk of acute tubular necrosis. Although the classic picture of
lower nephron nephrosis may not develop, a similar nonspecific acute renal
failure is noted in some cases of mercury (especially mercuric chloride)
poisoning and following exposure to radiocontrast agents, especially in
patients with diabetes mellitus or myeloma.
Clinical
Findings
A.
Symptoms and Signs: Usually the clinical
picture is that of the associated clinical state. Dehydration and shock may be
present concurrently, but the urine output and acute renal failure fail to
improve following administration of intravenous fluids, in contrast to the
situation in patients who have pre-renal renal failure (see preceding
discussion). On the other hand, there may be signs of excessive fluid retention
in patients with acute renal failure following radiocontrast exposure. Symptoms
of uremia per se (eg, altered mentation or gastrointestinal symptoms) are
unusual in acute renal failure (in contrast to chronic renal failure).

Cardiovascular. Hypertension, pulmonary
edema, peripheral edema, and arrhythmias are among the cardiovascular effects
of ARF. As decreased urine output causes fluid retention, the patient develops
hypertension. If his heart can’t pump the additional volume, heart failure
ensues. Renin overproduction also causes hypertension.
Edema results when extra fluid moves from his blood vessels into the
interstitial space or into his tissues. Because blood albumin is essential to
regulate the passage of water and solutes through the capillaries and to
prevent fluid from shifting into the interstitial spaces, a low serum albumin
level can lead to edema.
Hyperkalemia occurs when the kidneys fail to excrete excess potassium. Be
on guard for signs and symptoms such as muscle weakness, loss of muscle tone,
and neuromuscular irritability, including tingling in the lips or fingertips.
Changes in the electrocardiogram, such as a flattened p wave, prolonged QRS
complex, and tall, tented T waves, also signal hyperkalemia. Often, though,
hyperkalemia doesn’t cause symptoms, so monitoring the patient’s serum
potassium level is critical. A level above 6 mEq/liter
could trigger bradycardia, heart block, asystole, or another arrhythmia.
|
Glomerular filtration rate—the key to kidney function Glomerular filtration rate (GFR), the amount of blood filtered through
the glomeruli of the kidneys, is the driving force of urine production.
Normally 120 to 125 ml/minute, GFR decreases when renal insult occurs.
Increased blood urea nitrogen and creatinine levels are indirect indicators
of reduced GFR. |
Respiratory. If fluid
overload continues, your patient may develop dyspnea, indicating pulmonary
edema. Assess him for dyspnea at rest or on exertion and auscultate his lungs
for crackles.
 Pulmonary edema
Pulmonary edema
Neurologic.
Metabolic wastes building up in his blood can affect your patient’s mental
status. Look for changes in his level of consciousness, which could progress to
coma. Sensory changes and weakness in the extremities signal uremic neuropathy.
Hematologic.
Anemia is the main hematologic effect of ARF. Contributing factors include
impaired red blood cell (RBC) production, hemolysis, bleeding, hemodilution,
and reduced RBC survival. The normal life span of RBCs, about 120 days, is
shortened to about 60 days in ARF. And because the damaged kidneys produce less
and less erythropoietin to stimulate RBC production, the lost RBCs aren’t
replaced. Monitor your patient for decreased hemoglobin and hematocrit levels
and dyspnea due to insufficient oxygenation.
Gastrointestinal (GI). Uremia causes anorexia, nausea, and vomiting, which lead to poor nutrition
and loss of body mass and muscle. Because uremia also can trigger colitis and
gastric ulcers, your patient is at risk for GI bleeding. His breath may take on
a foul urine odor caused by an increase in urea.
To confirm ARF, the primary care provider will perform a thorough history
and physical examination, blood and urine tests, and possibly a renal
ultrasound. If testing confirms ARF, prepare to intervene rapidly to prevent
permanent renal damage.
Eliminating the cause
In most cases of ARF, normal kidney function returns naturally within
weeks. The treatment goals are to eliminate the cause of ARF and support the
patient’s kidney function and other affected body systems. This means
reestablishing blood flow to the kidneys for a prerenal condition, treating
intrinsic renal disease such as acute glomerulonephritis, or removing a
postrenal obstruction. Regardless of the type of ARF affecting your patient,
provide the following supportive measures:
Maintain fluid and electrolyte balance.
Accurately assessing fluid balance is critical, so strictly monitor your
patient’s weight along with his fluid intake and output. Be sure to include
vomitus and liquid stools in output measurements.
Assess him for edema, which is often dependent (in the legs and feet if
he’s sitting or in the sacral region if he’s supine) but also may appear around
his eyes. Document the color and clarity of his urine.
A patient with ARF has an increased risk of hyperkalemia, hyponatremia, and
volume overload, so closely monitor his fluid and electrolyte levels. A trial
of diuretic therapy may remove excess fluid and electrolytes.
B.
Laboratory Findings:
1. Urine-Although the specific gravity may be high
immediately after the acute event, it usually becomes low or fixed in the
1.005-1.015 range. Urine osmolality is also low (< 450 mOsm/kg and U/P
os-molal ratio < 1.5:1). Urinalysis often discloses tubular cells and
granular casts; the urine may be muddy brown. If the test for occult blood is
positive, one must be concerned about the presence of myoglobin as well as
hemoglobin. Tests for differentiating myoglobin pigment are available.
2. Central
venous pressure-This is usually normal to slightly elevated.
3. Fluid
challenges-There is no increase in urine volume following intravenous
administration of mannitol or physiologic saline. Occasionally following the
use of furosemide or "renal doses" of dopamine (1-5 µg/kg/min), a low
urine output is converted to a high fixed urine output (low-output renal
failure to high-output renal failure), but there is usually no change in the
rate of increase of blood urea nitrogen or creatinine.
Treatment
If
there is no response to the initial fluid or mannitol challenge, the volume of
administered fluid must be sharply curtailed and the amount given related to
measured fluid losses (eg, urine and gastrointestinal) and estimated
insensible fluid losses. An early assessment of the rate of rise of serum creatinine
and blood urea nitrogen and of the concentrations of electrolytes is necessary
to predict the possible use of dialysis therapy. There is some evidence that
early use of hyperalimentation might be beneficial in reducing both the need
for dialysis and the morbidity and mortality. With appropriate regulation of
the volume of fluid administered, solutions of glucose and essential amino
acids to provide 30-35 kcal/kg are used to correct or reduce the severity of
the catabolic state accompanying acute tubular necrosis.
Serum
or plasma potassium must be closely monitored and serial ECGs done to ensure
early-recognition of hyperkalemia. This condition can be treated with (1)
intravenous sodium bicarbonate administration, (2) Kayexalate, 25-50 g (with
sorbitol) orally or by enema, (3) intravenous glucose and insulin, and (4) intravenous
calcium preparations to prevent cardiac irritability.
Peritoneal
dialysis or hemodialysis should be used as necessary to avoid or correct
uremia, hypokalemia, or fluid overload. Hemodialysis in patients with acute
renal failure can be either intermittent or continuous (with arteriovenous or
venovenous hemofiltration techniques). Vascular access is obtained with percutaneous
catheters. The continuous dialysis techniques allow for easier management in
many hemodynami-cally unstable patients in intensive care units.
Prognosis
Most
cases are reversible within 7-14 days. Residual renal damage may be noted,
particularly in elderly patients.
POSTRENAL
ACUTE RENAL FAILURE

The
conditions listed in Table 1 involve primarily the need for urologic
diagnostic and therapeutic interventions. Following lower abdominal surgery,
urethral or ureteral obstruction should be considered as a cause of acute renal
failure. The causes of bilateral ureteral obstruction are (1) peritoneal or
retroperitoneal neoplastic involvement, with masses or nodes; (2)
retroperitoneal fibrosis; (3) calculous disease; and (4) postsurgical or
traumatic interruption. With a solitary kidney, ureteral stones can produce
total urinary tract obstruction and acute renal failure. Urethral or bladder
neck obstruction is a frequent cause of renal failure, especially in elderly
men.

A number of things can cause kidney failure, but
postrenal acute renal failure in particular is caused by anything that
interferes with the flow of urine out of your kidneys. All of the blood in your
body goes through the kidneys, where waste is usually filtered out and excreted
from the body as urine. However, in people with postrenal acute renal failure,
something interrupts the urine flow from the kidneys, which means waste can't
be filtered or disposed of correctly and waste builds up in the blood. Usually,
postrenal acute renal failure is caused by some kind of obstruction in the bladder
or its related passageways. Kidney stones or tumors in the ureters (the tubes
connecting your kidneys to your bladder) can block waste from passing into the
bladder. Also, anything that causes an obstruction in the bladder itself, such
as an enlarged prostate, a bladder stone, or a blood clot, may stop urine from
properly passing out of the kidneys and through the bladder.
Clinical
Findings

A. Symptoms
and Signs: Renal pain and renal tenderness often are present. If there has
been an operative ureteral injury with associated urine extravasation, urine
may leak through a wound. Edema from overhydration may be noted. Ileus is often
present along with associated abdominal distention and vomiting.
B. Laboratory
Findings: Urinalysis is usually not helpful. A large volume of urine
obtained by catheterization may be both diagnostic and therapeutic for lower
tract obstruction.
C. X-Ray
Findings: Poor visualization on intravenous urography is characteristic.
Radionuclide renal scans may show a urine leak or, in cases of obstruction,
retention of the isotope in the renal pelvis. Renal scans are helpful in acute
but not in chronic obstruction.
 KUB
(coral stoun in the right kidney)
KUB
(coral stoun in the right kidney)

Ultrasound examination often reveals a dilated upper collecting system with deformities
characteristic of hydronephrosis.
 CT
CT

D. Instrumental
Examination: Cystoscopy and retrograde
ureteral catheterization demonstrate ureteral obstruction.

 Cystoscopy
Cystoscopy
Treatment
Treatment options for
postrenal acute renal failure may vary depending on the cause of the condition.
In instances where the cause of kidney failure is something that can be treated
or removed, such as bladder obstruction or kidney stones, it's important to
treat that condition first. Once any underlying cause or obstruction has been
treated, the kidneys may be able to heal themselves. To promote kidney healing,
it's important to replace fluids and minerals that may have been lost. It's
also important to maintain a balanced diet that's high in carbohydrates and low
in potassium. If levels of certain minerals are too high, doctors may recommend
calcium salts or sodium polystyrene sulfonate to be taken, usually by mouth.
Doctors may also put you on short-term dialysis, which involves pumping your
blood through a machine that filters the blood and removes waste.
Treatment for end-stage kidney disease
If your kidneys can't keep up
with waste and fluid clearance on their own and you develop complete or
near-complete kidney failure, you have end-stage kidney disease. At that point,
dialysis or a kidney transplant is needed.
Dialysis.
Dialysis artificially removes waste products and extra fluid from your
blood when your kidneys aren't able to perform these functions. In
hemodialysis, a machine filters waste and excess fluids from your blood. In
peritoneal dialysis, you use a catheter to fill your abdominal cavity with a
dialysis solution that absorbs waste and excess fluids — then this solution
drains out of your body and is replaced with fresh solution.
Kidney transplant. 

If you have no life-threatening
medical conditions other than kidney failure, a kidney transplant may be an
option for you. Kidney transplant involves surgically placing a healthy kidney
from a donor into your body. Transplanted kidneys can come from deceased donors
or from living donors.


Urological Emergencies 
Overview
Some urological conditions have serious or
life-threatening consequences and require immediate medical attention. These
medical emergencies include
·
acute urinary retention,
·
Fournier’s gangrene,
·
paraphimosis,
·
priapism, and
·
testicular
torsion.
While these conditions are unrelated and have
different symptoms, they all require urgent care. Delaying treatment in some
cases can result in surgical removal of testicles (orchiectomy), permanent
inability to achieve an erection (impotence), or death.
Acute
urinary retention is the sudden inability to urinate and is usually symptomatic
of another condition that needs treatment.
Incidence and Prevalence
Anyone can experience acute urinary
retention. The causes and rate of occurrence varies greatly between genders
until about age 60, when men are more often affected as a result of benign prostatic hyperplasia (BPH).
 Benign prostatic hyperplasia (BPH).
Benign prostatic hyperplasia (BPH).
Risk Factors
Kidney stones, prostate cancer, prostatitis,
and BPH are risk factors in men. Women with a history of kidney stones or
urinary tract infections (UTIs), pregnant women, and those who have had recent
gynecological surgery are at higher risk.
Causes
Acute urinary retention is caused by obstruction in
the bladder or the tube that carries urine from the bladder outside the body
(urethra), a disruption of sensory information in the nervous system (e.g.,
spinal cord or nerve damage), or a situation or event that causes the bladder
to become distended.
Factors associated with acute urinary retention
include the following:
·
Alcohol consumption
·
Allergy or cold medications containing
decongestants or antihistamines
·
Certain prescription drugs (e.g.,
ipratropium bromide, albuterol, epinephrine) that cause the urethra to become
narrow
·
Delaying urination for a long time
·
Long period of inactivity or bed rest
·
Prolonged exposure to cold temperatures
·
Spinal cord injury/nerve damage
·
Surgery (e.g., complication of anesthesia)
·
Urinary system obstruction (e.g., benign
prostatic hyperplasia (BPH), kidney stones)
·
urinary tract infection
Signs and Symptoms
Acute urinary retention
produces severe lower abdominal pain, a distended abdomen, and/or the sudden
inability to pass urine.
Complications that may develop with
untreated urinary retention include bladder damage and chronic kidney failure.
Diagnosis
Diagnosis is based on a sudden lack of urinary output
and bladder swelling
(distention) observed during a physical examination. 

Treatment
Treatment should be obtained within 5 hours of the
onset of symptoms to avoid the development of complications. The underlying
cause of urinary retention (e.g., kidney stones) must be diagnosed and treated
as well.

 Catheterization of the bladder
Catheterization of the bladder
A small tube (catheter) is inserted into the bladder
through the urethra to drain the urine. Catheterization relieves pain and
distention.
Prognosis
Depending on the underlying cause, the recurrence rate can be up to 70%
within a week after initial treatment. BPH is responsible for most recurrences.
Fournier’s
Gangrene 
Fournier’s gangrene, sometimes called Fournier’s disease, is a bacterial
infection of the skin that affects the genitals and perineum (i.e., area
between the scrotum and anus in men and in women between the vulva and anus).
The disease develops after a wound or abrasion becomes infected. A combination
of anaerobic (living without oxygen) microorganisms (e.g., staphylococcal) and
fungi (e.g., yeast) causes an infection that spreads quickly and causes
destruction (necrosis) of skin, tissue under the skin (subcutaneous tissue),
and muscle. Staphylococcal bacteria clot the blood, depriving surrounding
tissue of oxygen. The anaerobic bacteria thrive in this oxygen-depleted
environment and produce molecules that instigate chemical reactions (enzymes)
that further the spread of the infection. Fournier’s gangrene can be fatal if
the infection enters the bloodstream.
Incidence
and Prevalence
Men are
ten times more likely than women to develop Fournier’s gangrene. Men aged 60-80
with a predisposing condition are most susceptible.
Women
who have had a pus-producing bacterial infection (abscess) in the vaginal area,
a surgical incision in the vagina and perineum to prevent tearing of skin
during delivery of a child (episiotomy), an abortion resulting in fever and an
infection of the lining of the uterus (septic abortion), or surgical removal of
the uterus (hysterectomy) are susceptible.
Rarely,
children may develop Fournier’s gangrene as a complication in wounds that
result from a burn, circumcision, or insect bite.
Risk
Factors
Men
with alcoholism, diabetes mellitus, leukemia, morbid obesity, and immune system
disorders (e.g., HIV, Crohn’s disease), and intravenous drug users are at
increased risk for developing Fournier’s gangrene. Surgery is also a risk
factor.
Causes
Fournier’s gangrene develops when multiple bacteria infect the body
through a wound, usually in the perineum, tube that carries urine outside the
body from the bladder (urethra), or colorectal area. Existing immune system
deficiencies help infection to spread quickly, producing a disease that
destroys the skin and superficial and deep fascia (membranes that separate
muscles and protect nerves and vessels) of the genital area. The chambers in
the penis that fill with blood to create an erection (corpora cavernosa),
testicles, and urethra are not usually affected.
Signs
and Symptoms
The early physical symptoms of Fournier’s gangrene do not always
indicate the severity of the condition. Pain sometimes diminishes as the
disease progresses. Symptoms are progressive and include the following:
·
Crepitant ("spongy" to the
touch) skin
·
Dead and discolored (gray-black) tissue;
pus weeping from injury
·
Fever and drowsiness (lethargy)
·
Increasing genital pain and redness
(erythema)
·
Odor
·
Severe genital pain accompanied by
tenderness and swelling of the penis and scrotum
Diagnosis 
Physical examination and blood tests are necessary. A diagnosis is made
on finding gangrenous (i.e., spongy, weeping, discolored) skin.

Microscopic examination of a tissue specimen (biopsy) may be taken if
visible symptoms are insufficient to distinguish between Fournier’s and other
bacterial infections.
Treatment
Antibiotics (often double or triple drug therapy)
along with aggressive surgical removal of all of the diseased tissue is
required immediately for an optimal outcome. 

Without early treatment, bacterial infection enters the bloodstream and
can cause delirium, heart attack, respiratory failure, and death.
Complications
Incomplete debridement (surgical removal of
dead tissue) allows wound infection to continue to spread. In this event,
follow-up surgery is performed.
Paraphimosis
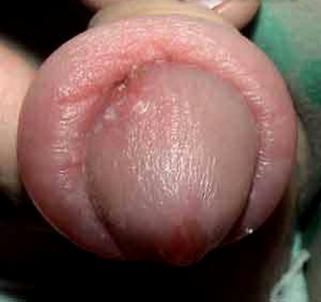
Paraphimosis occurs when the fold of skin that covers the head (glans)
of an uncircumcised penis (i.e., the foreskin) has been retracted and narrows
below the glans, constricting the lymphatic drainage and causing the glans to
swell. If not corrected, blood flow in the penis becomes impeded by the
increasingly constricting band of foreskin, which causes further swelling of
the glans. Because lack of oxygen from the reduced blood flow can cause tissue death
(necrosis), paraphimosis is considered a medical emergency and requires
immediate treatment.
Incidence
and Prevalence
In the
Risk
Factors
Uncircumcised males are at risk. Piercing the penis increases the risk
if the penile ring interferes with foreskin retraction or replacement over the
glans, and if infection results from the piercing.
Causes
 Paraphimosis
Paraphimosis
Causes include the following:
·
Bacterial infection (e.g.,
balanoposthitis)
 Balanoposthitis
Balanoposthitis
·
Catheterization (i.e., if the foreskin is
not returned to its original position after a urethral catheter is inserted,
the glans may become swollen, which can initiate paraphimosis)
·
Poor hygiene
·
Swelling-producing injury
·
Vigorous sexual intercourse
Signs and Symptoms
Symptoms include the following:
·
Band of retracted foreskin tissue beneath
the glans
·
Black tissue on the glans (indicates
necrosis)
·
Inability to urinate (urinary retention)
·
Penile pain
·
Redness (erythema)
·
Swollen glans (the shaft of the penis is
not swollen)
·
Tenderness
Complications
Tissue death caused by loss of blood supply (gangrene) and spontaneous
detachment of diseased tissue (autoamputation) of the glans are possible complications
of paraphimosis.
Diagnosis
Paraphimosis is diagnosed during a physical examination.
Treatment
Because paraphimosis can be severely painful, a pain reliever is
administered before treatment. The first method of treatment after diagnosis involves
manual manipulation of the penis to reduce swelling and to replace the foreskin
over the glans.
 Manual
manipulation of the penis
Manual
manipulation of the penis
An ice pack may be applied to the penis (after the penis has been
wrapped in plastic) to help reduce swelling.


If manual treatment is unsuccessful, the puncture technique uses
a needle to drain excess watery fluid in the swollen tissue (edematous fluid)
from the glans to reduce swelling.
A third option is to make a small incision in the foreskin to alleviate constriction
and allow the swelling to subside. With this procedure, local anesthesia is
administered to minimize discomfort.
After reduction of swelling is achieved, antibiotics are prescribed for
any underlying infection.
Prognosis
Full recovery from paraphimosis is expected with prompt treatment.
Prevention
Circumcision
is recommended after treatment to prevent a recurring episode.
Priapism
Priapism is a prolonged, painful penile erection that occurs when blood
in the penis is "trapped," or unable to drain. The stagnant blood
causes an erection that can last from hours to days. A painful erection lasting
for more than 4 hours indicates priapism. If not treated promptly, scarring and
permanent inability to achieve an erection (impotence) can result.
Types
Veno-occlusive
(low flow)
Veno-occlusive
(blocked vein) priapism develops when circulation in the penis becomes sluggish
due to obstructed veins. This type usually occurs without a known cause in men
who are otherwise healthy.
Arterial
(high flow)
This
rare, less painful type of priapism results from an injury to the penis or area
between scrotum and anus (perineum) that prevents blood in the penis from
circulating normally. It indicates a ruptured artery in the penis. There may be
a lapse between the time of injury and onset of priapism. Incidence and
Prevalence Priapism can affect men of any age. Most venoocclusive priapism in
men with sickle cell disease occurs between ages 19-21. The rate of
venoocclusive priapism is higher in men who have malaria, leukemia, and Fabry
disease.
Risk
Factors
Diseases that affect blood circulation may predispose men to developing
the condition. Forty-two percent of men with sickle cell disease develop
veno-occlusive priapism at least once.
Recreational or "party" drug use (e.g., cocaine, ecstasy,
marijuana) is a risk factor. An overdose of injectable medication such as
papaverine and phentolamine (Regitine®) for erectile dysfunction is also a risk
factor. Men with sickle cell disease, leukemia, malaria, and Fabry disease are
predisposed to priapism.
Alcohol consumption, androgenic steroids (used to increase muscle size),
anticoagulants (Coumadin®, Warfilone®), and antihypertensives (Prazosin®)
increase risk. Prolonged sexual activity is also a risk factor.
Causes
Priapism may develop as a result of prolonged sexual activity. Other
causes include the following:
·
Black widow spider bites
·
Carbon monoxide poisoning
·
Erectile dysfunction injection therapy (if
amount of medication injected exceeds prescribed dose)
·
Penile or perineal injury (e.g., perineal
trauma against the top tube of a bicycle)
·
Prescription antidepressive drugs
trazodone (Desyrel®) and chlorpromazine (Compazine®, Serentil®)
·
Spinal cord trauma
· Tumor
Signs
and Symptoms
A painful penile erection that lasts 4 hours or more, and a soft head
(glans) with a hard shaft are signs of priapism.
Diagnosis
Diagnosis includes a patient history and a physical examination to
detect an injury or underlying problem.
In veno-occlusive priapism, angiography may be used to help
locate blocked veins. Angiography uses a special dye injected into the
bloodstream to enable the physician to see blockages on x-ray.
Doppler sonogram (i.e., digital images of ultrasound echos that detect
poor blood flow) may be used to diagnose high- or low-flow priapism.
Treatment
There are several forms of treatment. Ice packs are applied to the penis
and perineum to reduce swelling. Walking up a flight of stairs is sometimes
effective, because mild exercise may divert blood flow to other areas of the
body. The underlying injury (i.e., ruptured artery) causing arterial priapism
is treated by tying off the artery (surgical ligation) to restore normal blood
flow.
Intracavernous injection 

Low-flow priapism is treated with vasoconstrictive medications injected
into the chambers in the penis that fill with blood to
create an erection (corpora cavernosa) to narrow the veins and cause swelling
to subside. Alpha agonists terbutaline (Adrenalin®,
Alupent®) and phenylephrine (Neo-Synephrine®) are commonly used.
Puncture

After numbing the area, a needle is used to
drain the blood from the corpora cavernosa to allow the swelling to subside.
Surgical
shunt 
For veno-occlusive priapism, a passageway (shunt) may be surgically
inserted to divert blood flow and reestablish circulation.
The underlying cause is treated when disease is present (e.g., leukemia,
sickle cell disease).
Prognosis
The prognosis is good for both types of priapism when the condition is resolved
quickly. When treatment is delayed, penile scarring and permanent impotence can
result.
Testicular
Torsion

Testicular torsion is a disorder in which the testicles rotate (twist) and
strangle the spermatic cord, which consists of blood vessels, lymphatic
vessels, nerves, and the duct that carries sperm from the body (vas deferens),
cutting off the blood supply to the testicles. Torsion can cause shrinkage
(atrophy) and tissue death (necrosis), and may require surgical removal of the
testicles (orchiectomy) if not treated promptly. Torsion often occurs during
sleep.
 Testicular
torsion
Testicular
torsion
Incidence
and Prevalence
Testicular
torsion primarily affects infants in the first year of life and adolescent boys
age 12-18, although it can occur at any age. Males with one or both testicles
not descended into scrotum (cryptorchidism) develop testicular torsion more
often than the general population.
Risk
Factors
Injury to the scrotum or groin and vigorous physical activity are risk
factors.

Causes
Injury to the scrotum can initiate a muscle spasm that
cause the testicles to twist. Some cases result from inadequate
connective tissue that "anchors" the testicle within the scrotum.
Many cases are idiopathic (i.e., have no known cause).
Signs
and Symptoms
Symptoms include the following:
·
Blood in semen
·
Lower abdominal pain
·
Lump in testicle
·
Nausea and vomiting
·
Sudden, severe testicular pain, followed
by diminishing pain after several hours (after necrosis begins to set in)
·
Redness of scrotum
·
Swelling of one testicle
Diagnosis
A patient history and physical examination is usually sufficient to
diagnose testicular torsion. Testicular torsion may cause symptoms (e.g.,
testicular pain and swelling) similar to epididymitis (i.e., inflammation of
the tubule where sperm is stored) and diagnostic tests may be necessary.
Color Doppler sonography (color printout of an ultrasound
echo test) is used to identify the absence of blood flow typically found in a
twisted testicle, which distinguishes the condition from epididymitis.
Urinalysis (analyzing chemical composition of urine)
can be used to rule out bacterial infections.
Surgical exploration may be necessary if diagnosis cannot be made using
other methods.
Treatment
Treatment involves untwisting (detorsion), manually if possible and
surgically if necessary. Surgical detorsion requires anesthesia followed by an
incision in the scrotum.


The testicles are untwisted and evaluated for necrosis. Dead tissue is
removed; removal of one or both testicles may be necessary. If necrosis has not
occurred, the healthy testicle(s) are then sutured (stitched) to the scrotal
wall to avoid recurrence.
 necrosis
necrosis
Prognosis
If torsion is diagnosed and treated within 5-6 hours, the prognosis is good.
The more time that elapses before resolution worsens the prognosis. After 18-24
hours, necrosis usually develops and indicates removal of the affected testicle
INJURIES TO THE BLADDER
Bladder
injuries occur most often from external force and are often associated with
pelvic fractures. (About 15% of all pelvic fractures are associated with
concomitant bladder or urethral injuries.) latrogenic
injury may result from gynecologic and other extensive pelvic procedures as
well as from hernia repairs and transurethral operations.
Pathogenesis & Pathology
The
bony pelvis protects the urinary bladder very well. When the pelvis is
fractured by blunt trauma, fragments from the fracture site may perforate the
bladder. These perforations usually result in extraperitoneal rupture. If the
urine is infected, extraperitoneal bladder perforations may result in deep
pelvic abscess and severe pelvic inflammation.
 Intraperitoneal
disruption
Intraperitoneal
disruption
When the bladder is filled to near capacity, a direct blow to the lower
abdomen may result in bladder disruption. This type of disruption ordinarily is
intraperitoneal. Since the reflection of the pelvic peritoneum covers the dome
of the bladder, a linear laceration will allow urine to flow into the
abdominal cavity. If the diagnosis is not established immediately and if the
urine is sterile, no symptoms may be noted for several days. If the urine is
infected, immediate peritonitis and acute abdomen will develop.
Clinical Findings
Pelvic
fracture accompanies bladder rupture in 90% of cases. The diagnosis of pelvic
fracture can be made initially in the emergency room by lateral compression on
the bony pelvis, since the fracture site will show crepitus and be painful to
the touch. Lower abdominal and suprapubic tenderness is usually present.
Pelvic fracture and suprapubic tenderness with acute abdomen suggest
intraperitoneal bladder disruption.
A. Symptoms: There is usually a history of lower abdominal trauma. Blunt injury is
the usual cause. Patients ordinarily are unable to urinate, but when
spontaneous voiding occurs, gross hematuria is usually present. Most patients
complain of pelvic or lower abdominal pain.
B. Signs:
Heavy bleeding associated with pelvic fracture may result in hemorrhagic
shock, usually from venous disruption of pelvic vessels. Evidence of external
injury from a gunshot or stab wound in the lower abdomen should make one suspect
bladder injury, manifested by marked tenderness of the suprapubic area and
lower abdomen. An acute abdomen indicates intraperitoneal bladder rupture. A
palpable mass in the lower abdomen usually represents a large pelvic hematoma.
On rectal examination, landmarks may be indistinct because of a large pelvic
hematoma.
C. Laboratory Findings: Catheterization
usually is required in patients with pelvic trauma but not if bloody urethral
discharge is noted. Bloody urethral discharge indicates urethral injury, and a urethrogram is necessary before catheterization. When
catheterization is done, gross or, less commonly, microscopic hematuria is
usually present. Urine taken from the bladder at the initial catheterization
should be cultured to determine whether infection is present.
D. X-Ray
Findings: A plain abdominal film generally demonstrates pelvic fractures. There
may be haziness over the lower abdomen from blood and urine extravasation. An
intravenous urogram should be obtained to establish whether kidney and ureteral
injuries are present.
 Retrogradual
cystography (Retroperitoneal rupture)
Retrogradual
cystography (Retroperitoneal rupture)
Bladder
disruption is shown on cystography. The bladder should be filled with 300 mL of
contrast material and a plain film of the lower abdomen obtained. Contrast
medium should be allowed to drain out completely, and a second film of the
abdomen should be obtained. The drainage film is extremely important, because
it demonstrates areas of extraperitoneal extravasation of blood and urine that
may not appear on the filling film. With intraperitoneal extravasation, free
contrast medium is visualized in the abdomen, highlighting bowel loops.
 Retrogradual
cystography (Intraperitoneal
rupture)
Retrogradual
cystography (Intraperitoneal
rupture)

 Retrogradual
cystography (Intraperitoneal
rupture)
Retrogradual
cystography (Intraperitoneal
rupture)
CT
cystography is an excellent method for detecting bladder rupture; however,
retrograde filling of the bladder with 300 mL of contrast medium is necessary
to distend the bladder completely. Incomplete distention with consequent missed
diagnosis of bladder rupture often occurs when the urethral catheter is
clamped during standard abdominal CT scan with intravenous contrast injection.
E.
Instrumental Examination: If urethral injury is suspected (bloody
discharge), a urethrogram should be obtained before
any attempt is made to catheterize the patient. If there is no evidence of urethral
injury, catheterization can be safely accomplished.
Cystoscopy
is not indicated, since bleeding and clots obscure visualization and prevent
accurate diagnosis.
Differential Diagnosis
Abdominal
trauma with hematuria may cause injury to the kidney and ureter as well as the
bladder. A urogram is indicated for all patients with trauma-related
hematuria. Associated injuries to the pelvic vessels and bowel also should be
considered.
The
urethra may be injured as well as the bladder; this possibility should be
considered in any patient with blunt trauma and pelvic fractures.
Urethrogra-phy demonstrates disruption of the urethra.
Complications
A
pelvic abscess may develop from extraperi-toneal bladder rupture; if the urine
becomes infected, the pelvic hematoma becomes infected too.
Intraperitoneal
bladder rupture with extravasation of urine into the abdominal cavity causes
delayed peritonitis.
Partial
incontinence may result from bladder injury when the laceration extends into
the bladder neck. Meticulous repair may ensure normal urinary control.
Treatment
A. Emergency
Measures: Shock and hemorrhage should be treated.
B. Surgical
Measures: A lower midline abdominal incision should be made.
 Suprapubic cystostomy
Suprapubic cystostomy
As the bladder is approached in the midline, a
pelvic hematoma, which is usually lateral, should be
avoided. Entering the pelvic hematoma can result in increased bleeding from
release of tamponade and in infection of the hematoma, with subsequent pelvic
abscess. The bladder should be opened in the midline and carefully inspected.
After repair, a suprapubic cystostomy tube is usually left in place to ensure
complete urinary drainage and control of bleeding.
1.
Extraperitoneal rupture- Extraperitoneal rupture
should be repaired intravesically. As the bladder is opened in the midline, it
should be carefully inspected and lacerations closed from within. Polyglycolic
acid or chromic absorbable sutures should be used.
 Juncture by two-row catgut junctures.
Juncture by two-row catgut junctures.
 Perivesical space is drainage through
obturatorial foramen or ischiorectal space.
Perivesical space is drainage through
obturatorial foramen or ischiorectal space.
 Drainage through obturatorial foramen
Drainage through obturatorial foramen
Extraperitoneal
bladder lacerations occasionally extend into the bladder neck and should be
repaired meticulously. Fine absorbable sutures should be used to ensure
complete reconstruction so that the patient will have urinary control after
injury. Such injuries are best managed with indwelling urethral catheterization
and suprapubic diversion.
Peritoneotomy
should be done and the intraabdominal fluid inspected before the procedure is
completed. If abdominal fluid is bloody, complete abdominal exploration should
be done to rule out associated injuries.
2.
Intraperitoneal rupture Intraperitoneal bladder ruptures should
be repaired via a transperitoneal approach after careful transvesical
inspection and closure of any other perforations.
 Transperitoneal
approach
Transperitoneal
approach
The
peritoneum must be closed carefully over the area of injury. The bladder is
then closed in separate layers by absorbable suture. All extravasated fluid
from the peritoneal cavity should be removed before closure. At the time of closure,
care should be taken that the suprapubic cystostomy is in the extraperitoneal
position.
3. Pelvic fracture Stable
fracture of the pubic rami is usually present. In such cases, the patient can
be ambulatory within 4-5 days without damage or difficulty. Unstable pelvic
fractures requiring external fixation have a more protracted course.
4.
Pelvic hematoma There may be heavy uncontrolled bleeding from rupture
of pelvic vessels even if the hematoma has not been entered at operation. At
exploration and bladder repair, packing the pelvis with laparotomy tapes often
controls the problem. If bleeding persists, it may be necessary to leave the
tapes in place for 24 h and operate again to remove them. Embolization of
pelvic vessels with Gelfoam or skeletal muscle under angiographic control is
useful in controlling persistent pelvic bleeding.
C.
Medical Measures: The
patient whose cystogram shows only a small degree of extravasation can be
managed by placing a urethral catheter into the bladder, without operation or
suprapubic cystostomy. The urine must be free of infection. Corriere and
Sandier (1988) have reported success with such management. Careful observation
is necessary because of the potential for pelvic hematoma infection, continued
bleeding from the bladder, and clot retention.
Prognosis
With
appropriate treatment, the prognosis is excellent. The suprapubic cystostomy
tube can be removed within 10 days, and the patient can usually void normally.
Patients with lacerations extending into the bladder neck area may be
temporarily incontinent, but full control is usually regained. At the time of
discharge, urine culture should be performed to determine whether
catheterassociated infection requires further treatment.
INJURIES TO THE URETHRA

Urethral
injuries are uncommon and occur most often in men, usually associated with
pelvic fractures or straddle-type falls. They are rare in women.
Various
parts of the urethra may be lacerated, transected, or contused. Management
varies according to the level of injury. The urethra can be separated into 2
broad anatomic divisions: the posterior urethra, consisting of the prostatic
and membranous portions, and the anterior urethra, consisting of the bulbous
and pendulous portions.
INJURIES TO THE POSTERIOR URETHRA
Etiology
The
membranous urethra passes through the urogenital diaphragm and is the portion
of the posterior urethra most likely to be injured. The urogenital diaphragm
contains most of the voluntary urinary sphincter. It is attached to the pubic
rami inferiorly, and when pelvic fractures occur from blunt trauma, the
membranous urethra is sheared from the prostatic apex at the prostatomembranous
junction. The urethra can be transected by the same mechanism at the interior
surface of the membranous urethra.
Pathogenesis & Pathology
Injuries
to the posterior urethra commonly occur from blunt trauma and pelvic fractures.
The urethra usually is sheared off just proximal to the urogenital diaphragm,
and the prostate is displaced superiorly by the developing hematoma in the
periprostatic and perivesical spaces.
Clinical Findings
A. Symptoms: Patients usually
complain of lower abdominal pain and inability to urinate. A history of
crushing injury to the pelvis is usually obtained.
B. Signs:
Blood at the urethral meatus is the single most important sign of urethral
injury. The importance of this finding cannot be overemphasized, because an
attempt to pass a urethral catheter may result in infection of the
periprostatic and perivesical hematoma and conversion of an incomplete
laceration to a complete one. The presence of blood at the external urethral
meatus indicates that immediate urethrography is necessary to establish the
diagnosis.
Suprapubic
tenderness and the presence of pelvic fracture are noted on physical
examination. A large developing pelvic hematoma may be palpated. Peri-neal or
suprapubic contusions are often noted. Rectal examination may reveal a large
pelvic hematoma with the prostate displaced superiorly. Rectal examination can
be misleading, however, because a tense pelvic hematoma may resemble the
prostate on palpation. Superior displacement of the prostate does not occur if
the puboprostatic ligaments remain intact. Partial disruption of the
membranous urethra (currently 10% of cases) is not accompanied by prostatic
displacement.
C. Laboratory
Findings: Anemia due to hemorrhage may be noted. Urine usually cannot be
obtained initially, since the patient should not void and catheterization
should not be attempted.
D. X-Ray
Findings: Fractures of the bony pelvis are usually present. A urethrogram (using 20-30 mL of watersoluble contrast
material) shows the site of extravasation at the prostatomembranous junction.

 Urethrogram
Urethrogram
(free extravasation of contrast material)
Ordinarily,
there is free extravasation of contrast material into the perivesical space.
Incomplete prostatomembranous disruption is seen as minor extravasation, with
a portion of contrast material passing into the prostatic urethra and bladder.
E. Instrumental
Examination: The only instrumentation involved should be for
urethrography. Catheterization or urethroscopy should not be done, because
these procedures pose an increased risk of hematoma, infection, and further
damage to partial urethral disruptions.
Differential Diagnosis
Bladder
rupture may be associated with posterior urethral injuries. An intravenous
urogram should be considered part of the assessment. Delayed films should be
obtained to demonstrate the bladder and note extravasation. Cystography cannot
be done preoperatively, since a urethral catheter should not be passed. Careful
evaluation of the bladder at operation is necessary. The anterior portion of
the urethra may be injured as well as the prostatomembranous urethra.
Complications
Stricture,
impotence, and incontinence as complications of prostatomembranous disruption
are among the most severe and debilitating mishaps that result from trauma to
the urinary system. Stricture following primary repair and anastomosis occurs
in about one-half of cases. If the preferred suprapubic cystostomy approach
with delayed repair is used, the incidence of stricture can be reduced to
about 5%.
The
incidence of impotence after primary repair is 30-80% (mean, about 50%). This
figure can be reduced to 10-15% by suprapubic drainage with delayed urethral
reconstruction.
Incontinence
in primary reanastomosis is noted in one-third of patients. Delayed
reconstruction reduces the incidence to less than 5%.
Treatment
A. Emergency
Measures: Shock and hemorrhage should be treated.
B. Surgical
Measures: Urethral catheterization should be avoided.
1.
Immediate management-Initial management
should consist of suprapubic cystostomy to provide urinary drainage.
 Suprapubic
cystostomy
Suprapubic
cystostomy
A
midline lower abdominal incision should be made, care being taken to avoid the
large pelvic hematoma. The bladder and prostate are usually elevated superiorly
by large periprostatic and perivesical hematomas. The bladder often is
distended by a large volume of urine accumulated during the period of
resuscitation and operative preparation. The urine is often clear and free of
blood, but gross hematuria may be present. The bladder should be opened in the
midline and carefully inspected for lacerations. If a laceration is present,
the bladder should be closed with absorbable suture material and a cystostomy
tube inserted for urinary drainage. This approach involves no urethral
instrumentation or manipulation. The suprapubic cystostomy is maintained in
place for about 3 months. This allows resolution of the pelvic hematoma, and
the prostate and bladder will slowly return to their anatomic positions.
Incomplete
laceration of the posterior urethra heals spontaneously, and the suprapubic
cystostomy can be removed within 2-3 weeks. The cystostomy tube should not be
removed before voiding cystourethrography shows that no extravasation persists.
2.
Urethral reconstruction-
 Urethral reconstruction
Urethral reconstruction


Urethral reconstruction
Reconstruction of the urethra
after prostatic disruption can be undertaken within 3 months, assuming there
is no pelvic abscess or other evidence of persistent pelvic infection. Before
reconstruction, a combined cystogram and urethrogram should be done to
determine the exact length of the resulting urethral stricture. This stricture
usually is 1-2 cm long and situated immediately posterior to the pubic bone.
The preferred approach is a single-stage reconstruction of the urethral
rupture defect with direct excision of the strictured area and anastomosis of
the bulbous urethra directly to the apex of the prostate. A 16F silicone
urethral catheter should be left in place along with a suprapubic cystostomy.
Catheters are removed within a month, and the patient is then able to void.
 Urethral reconstruction prostatomembranous part of the urethra
Urethral reconstruction prostatomembranous part of the urethra
3.
Immediate urethral realignment-Some surgeons
prefer to realign the urethra immediately. Direct suture reconstruction of the
prostatomembranous disruption in the acute injury is extremely difficult.
Persistent bleeding and surrounding hematoma create technical problems. The
incidence of stricture, impotence, and incontinence appears to be higher than
with immediate cystostomy and delayed reconstruction. However, several authors
have reported success with immediate urethral realignment.
C. General
Measures: After delayed reconstruction by a perineal approach, patients
are allowed ambulation on the first postoperative day and usually can be
discharged within 3 days.
D. Treatment
of Complications: Approximately 1 month after the delayed reconstruction,
the urethral catheter can be removed and a voiding cystogram obtained through
the suprapubic cystostomy tube. If the cystogram shows a patent area of reconstruction
free of extravasation, the suprapubic catheter can be removed; if there is
extravasation or stricture, suprapubic cystostomy should be maintained. A
follow-up urethrogram should be obtained within 2 months to watch for stricture
development.
Stricture,
if present (< 5%), is usually very short, and urethrotomy under direct
vision offers easy and rapid cure.
The
patient may be impotent for several months after delayed repair. Impotence is
permanent in about 10% of patients. Implantation of a penile prosthesis is
indicated if impotence is still present 2 years after reconstruction.
Incontinence
seldom follows transpubic or perineal reconstruction. If present, it usually
resolves slowly.
Prognosis
If
complications can be avoided, the prognosis is excellent. Urinary infections
ultimately resolve with appropriate management.
INJURIES TO THE ANTERIOR URETHRA
Etiology
The anterior urethra is the portion distal to
the urogenital diaphragm. Straddle injury may cause laceration or contusion of
the urethra. Self-instrumentation or iatrogenic instrumentation may cause
partial disruption.
Pathogenesis & Pathology
A. Contusion:
Contusion of the urethra is a sign of
crush injury without urethral disruption. Perineal hematoma usually resolves
without complications.
B. Laceration:
A severe straddle injury may result in laceration of part of the urethral
wall, allowing extravasation of urine. If the extravasation is unrecognized,
it may extend into the scrotum, along the penile shaft, and up to the abdominal
wall. It is limited only by Colics' fascia and often results in sepsis,
infection, and serious morbidity.
Clinical Findings
A. Symptoms:
There is usually a history of a fall, and in some cases a history of
instrumentation. Bleeding from the urethra is usually present. There is local
pain into the perineum and sometimes massive perineal hematoma. If voiding has
occurred and extravasation is noted, sudden swelling in the area will be
present. If diagnosis has been delayed, sepsis and severe infection may be
present.
B. Signs:
The perineum is very tender, and a mass may be found. Rectal examination
reveals a normal prostate. The patient usually has a desire to void, but
voiding should not be allowed until assessment of the urethra is complete. No
attempt should be made to pass a urethral catheter, but if the patient's
bladder is overdistended, percutaneous suprapubic cystostomy can be done as a
temporary procedure.
When
presentation of such injuries is delayed, there is massive urinary
extravasation and infection in the perineum and the scrotum. The lower abdominal
wall may also be involved. The skin is usually swollen and discolored.
C. Laboratory
Findings: Blood loss is not usually excessive, particularly if secondary
injury has occurred. The white count may be elevated with infection.
D. X-Ray
Findings: A urethrogram, with instillation of
15-20 mL of water-soluble contrast material, demonstrates extravasation and
the location of injury.
 Urethrogram
(demonstrates extravasation and the location of injury)
Urethrogram
(demonstrates extravasation and the location of injury)
A
contused urethra shows no evidence of extravasation.
E.
Instrumental Examination: If there is no evidence of extravasation
on the urethrogram, a urethral catheter may be passed into the bladder. Extravasation
is a contraindication to further instrumentation at this time.
Differential Diagnosis
Partial
or complete disruption of the prostatomem-branous urethra may occur if pelvic
fracture is present. Urethrography usually demonstrates the location and
extent of extravasation and its relationship to the urogenital diaphragm.
Complications
Heavy
bleeding from the corpus spongiosum injury may occur in the perineum as well
as through the urethral meatus. Pressure applied to the perineum over the site
of the injury usually controls bleeding. If hemorrhage cannot be controlled,
immediate operation is required.
The
complications of urinary extravasation are chiefly sepsis and infection.
Aggressive debridement and drainage are required if there is infection.
Stricture
at the site of injury is a common complication, but surgical reconstruction
may not be required unless the stricture significantly reduces urinary flow
rates.
Treatment
A. General
Measures: Major blood loss usually does not occur from straddle injury. If
heavy bleeding does occur, local pressure for control, followed by
resuscitation, is required.
B. Specific
Measures:
1. Urethral
contusion The patient
with urethral contusion shows no evidence of extravasation, and the urethra
remains intact. After urethrography, the patient is allowed to void; and if the
voiding occurs normally, without pain or bleeding, no additional treatment is
necessary. If bleeding persists, urethral catheter drainage can be done.
 Urethral catheter
Urethral catheter
2. Urethral
lacerations. Instrumentation of
the urethra following urethrography should be avoided. A small midline incision
in the suprapubic area readily exposes the dome of the bladder so that a
suprapubic cystostomy tube can be inserted, allowing complete urinary diversion
while the urethral laceration heals.
 PERCUTANEOUS CYSTOSTOMY
PERCUTANEOUS CYSTOSTOMY

PERCUTANEOUS CYSTOSTOMY
Percutaneous
cystostomy may also be used in such injuries. If only minor extravasation is
noted on the urethrogram, a voiding study can be performed within 7 days after
suprapubic catheter drainage to search for extravasation. In more extensive
injuries, one should wait 2-3 weeks before doing a voiding study through the
suprapubic catheter. Healing at the site of injury may result in stricture
formation. Most of these strictures are not severe and do not require surgical
reconstruction. The suprapubic cystostomy catheter may be removed if no extravasation
is documented. Follow-up with documentation of urinary flow rates will show
whether there is urethral obstruction from stricture.
3. Urethral
laceration with extensive urinary extravasation- After major laceration, urinary extravasation may involve
the perineum, scrotum, and lower abdomen. Drainage of these areas is indicated.
Suprapubic cystostomy for urinary diversion is required. Infection and abscess
formation are common and require antibiotic therapy.
4. Immediate
repair-Immediate repair of urethral lacerations can be performed, but the
procedure is difficult and the incidence of associated stricture is high.
C.
Treatment of Complications: Strictures at the site of injury may be
extensive and require delayed reconstruction.
Prognosis
Urethral
stricture is a major complication but in most cases does not require surgical
reconstruction. If, when stricture resolves, urinary flow rates are poor and
urinary infection and urethral fistula are present, reconstruction is required.
Renal colic 
Renal colic is a type
of abdominal pain commonly caused by kidney stones.
What are the symptoms of renal colic?
Symptoms of renal colic are
related to the formation of crystals in the kidneys. These stones can block
urine output, causing pain, swelling and infection.
Clinical features 
Patients usually present with severe loin pain which may come suddenly
or slowly increase in severity. The pain is caused by stretching of the renal
pelvis and capsule as the stone obstructs the drainage of urine and by
overactive contractions of the ureter as it attempts to dislodge the
obstructing stone passing through it. Pain can be felt anywhere in the region
of the kidney and along the course of the ureter, commonly described as ‘loin
to groin pain’. 
Loin pain usually represents more
proximal obstruction with the pain moving more anteriorly and to the groin if
and when the stone moves further down the ureter. 
As the innervation of the testicle and the kidney is the same, pain may
also be referred to the testicle or labia. Other common symptoms include nausea
and vomiting, haematuria and lower urinary tract symptoms (usually irritative)
either due to a urinary tract infection or as a result of inflammation of the
bladder wall because of a stone lodged at the ureteral orifice. Patients may
also describe passing a stone or stone debris when
they micturate and when possible these should be kept for analysis. A good
history will often reveal predisposing and precipitant factors such as
intensive exercise, high protein intake, periods of immobility, previous
episodes, familial tendency and conditions associated with stone formation such
as recurrent urinary tract infection (UTI), inflammatory bowel disease,
metabolic disorders (including gout), kidney disease and endocrine disorders.
Examination 
Unlike a patient with peritonism, who characteristically lies still, a
renal colic sufferer will often be more fidgety as they try to find a more
comfortable position. Simple observations will often reveal the patient to be
tachycardic, usually due to the pain, but beware of a febrile patient who may
be showing signs of septic shock. Palpation of the abdomen may reveal
tenderness in the areas described above.
Previous Section
Next Section
Investigations
Patients with renal colic may undergo several investigations and the
timing of each varies depending on the time of presentation.
Urinalysis
Dipstick urine testing is readily available to all GPs and is normally
the first test to be performed. It will show the presence of blood in 85% of
patients with renal colic. Although the absence of blood should alert you to
other causes of the pain, it does not exclude renal colic .
The presence of nitrites and/or leucocytes obviously raises the suspicion of a
UTI and urine should be sent for microscopy and culture. Normal urine is acidic
with a pH around 5.5, a urinary pH greater than 7
raises the possibility of infection with urea splitting bacteria and the
presence of struvite stones. Alternatively, a pH of 5.4 or less might suggest
increased levels of uric acid in the urine and the risk of a uric acid stone
(uric acid precipitates at pH 5.4).
Further investigations on the urine, usually via a specialist ‘stone
clinic’, will include 24-hour urine collections to measure calcium, phosphate,
uric acid and creatinine clearance. In addition, a urine spot test will be
performed to detect cystine.
Differential diagnosis of
renal colic
Pancreatitis
Pyelonephritis
Hepatobiliary conditions, for example cholecystitis
Appendicitis
Abdominal aortic aneurysm
Inflammatory bowel disease
Bowel malignancy
Ectopic pregnancy
Ruptured ovarian cysts
Pleurisy, for example lower lobe pneumonia
Stone analysis
Quantitative infrared analysis (spectrophotometry) of the stone material
will reveal its mineral composition. Patients should be encouraged to collect
and keep all stone material that is passed.
Imaging
Patients will invariably have some form of imaging either in the initial
acute period after a referral to the emergency department or in the week
following a resolved episode of colic to ensure there is no ongoing
obstruction, which depending on the local arrangement, may well be the remit of
the GP. Imaging investigations include:
Plain
X-ray/intravenous urogram 
A plain X-ray of the kidneys, ureter and bladder will often reveal a
stone if it contains calcium. For an intravenous urogram, the plain film is
followed by the administration of intravenous contrast (assuming there is good
renal function)

and repeated X-rays at 30-minute intervals are
taken until the contrast is seen to have cleared from the unobstructed side.
The contrast will delineate the renal tract and demonstrate ureteric
obstruction, which is seen as hydronephrosis and ureteric dilatation above an
obstruction.
Computed
tomography 
Non-contrast spiral computed tomography (CT) has the highest degree of
diagnostic accuracy and its use is becoming more widespread. Out of hours,
however, the availability of a CT scanner with the skilled interpretation
needed somewhat limits its use.
Ultrasound 

Although detecting small stones on ultrasound is difficult, it can be
very useful in detecting hydronephrosis, which indicates some degree of
obstruction. Ultrasound is often more readily available and is also useful,
alongside CT, if there is some diagnostic uncertainty as the
radiologist/radiographer will usually be able to screen for an alternative
pathology. Magnetic resonance imaging is useful but rarely used as it is time
consuming and offers no significant benefit over spiral CT.
Management
Once a diagnosis has been made, which is usually apparent from the
history, prompt treatment needs to be given. Analgesia should be the first
priority. If, in the general practice setting, the patient cannot be attended
to within half an hour, then they should be referred to the local accident and
emergency department. This would ordinarily be via an ambulance as a paramedic
would be able to give strong analgesia. Non-steroidal anti-inflammatories
(NSAIDs), most commonly in the form of intramuscular (IM) or rectal (PR)
diclofenac, are the most effective. Subcutaneous or IM opioids (diamorphine,
morphine or pethidine) are less effective alternatives when NSAIDs are
contraindicated. Antiemetics should also be given,
especially if nausea and vomiting is severe or opioid analgesia is used.
The patient should be referred if there is any diagnostic uncertainty as
conditions that require immediate intervention often present in a similar
fashion to renal colic. Patients with persistent pain, who appear systemically
unwell or who have symptoms and signs of infection, should all be referred
urgently, as should patients who are anuric, significantly dehydrated or have a
solitary or transplanted kidney. Urgent drainage using either a ureteric stent
or by means of a nephrostomy is required if the upper urinary tract is both
obstructed and infected, as septicaemia (usually with a gram-negative organism)
is a very real and life-threatening risk. Similarly, emergency drainage or
removal of an obstructing stone is required if renal function deteriorates, the
patient becomes anuric, has intractable pain or vomiting or there is
obstruction in a solitary or transplanted kidney .
Surgical treatment options
Endoscopic
laser lithotripsy 
The endoscopic management of renal stones requires a short hospital
stay. Under a general anaesthetic, a ureteroscope is
passed retrogradely into the ureter until the obstructing stone is identified.
The stone can then either be retrieved using a collapsible wire basket or
broken up in situ using a laser or ballistic (hydraulic and mechanical)
lithotriptor. 
The use of baskets and ballistic lithotriptors can be troublesome, and
lithotripsy with modern lasers, such as the Holmium: YAG (Ho:YAG)
laser, is now the mainstay of treatment. Endoscopic laser lithotripsy has a
success rate of around 90% and in combination with extracorporeal shock wave
lithotripsy (ESWL) has virtually removed the need for open stone surgery. The
Ho:YAG laser works by producing a vaporization bubble
at the tip of the fibre-optic cable while in water. Although all lasers have
the advantage that they can be delivered via fibre optics, making them ideal
for use with flexible ureteroscopes, only modern lasers are able to deliver
enough localized energy to break up all types of stone into small enough
fragments for routine use. Complication rates for Ho:YAG
laser lithotripsy are low at around 4%. Complications may relate to the
endoscopy itself (trauma) or residual stone fragments that are themselves large enough to cause colicky symptoms. Localized
tissue scarring and fibrosis can occur, leading to ureteral strictures, but
this is rare and a rate comparable to non-laser treatments.
Extracorporeal
shock wave lithotripsy 
ESWL is a non-invasive outpatient procedure that uses focused shock
waves to break stones into smaller fragments, preferably less than

Shock waves, usually produced by
an electromagnet, are propagated through the body with little or no adverse
effect because there is a minimal difference in the density of soft tissues
they pass through. However, at a stone-fluid interface, there is a large
difference in density at which focused shock waves dissipate a large enough
amount of energy to break up the stone. Overall, ESWL achieves stone-free rates
approaching 80%, but this diminishes with large (more than
Percutaneous
nephrolithotomy 

Percutaneous
nephrolithotomy (PCNL) is an inpatient procedure performed under
general anaesthesia. A nephroscope is passed via an incision in the loin into
the collecting system.

Through this, the stone can be broken up and removed using either a
laser or mechanical lithotriptor. It is used when stones are not amenable to an
endoscopic approach or ESWL. UTI and catheter problems are fairly common after
PCNL but more serious complications including bleeding, septicaemia and
pneumothorax are thankfully rare.
Open
surgery 

If the above measures fail, then open surgery is required but this is
now an uncommon occurrence (1–5% of patients).
