Digestion of
proteins. General pathways of amino acids
transformation.
Proteins are essential
nutrients for the human body. They are one of the building blocks of body
tissue, and can also serve as a fuel source. As fuel, proteins contain 4 kcal
per gram, just like carbohydrates and unlike lipids, which contain 9
kcal per gram.
Proteins are polymer chains made of amino acids
linked together by peptide bonds. In nutrition, proteins
are broken down in the stomach during digestion
by enzymes
known as proteases
into smaller polypeptides to provide amino acids for the
body, including the essential amino acids that cannot be biosynthesized
by the body itself.
Amino acids can be divided into three
categories: essential amino acids, non-essential amino acids and conditional
amino acids. Essential amino acids cannot be made by the body, and must be
supplied by food. Non-essential amino acids are made by the body from essential
amino acids or in the normal breakdown of proteins. Conditional amino acids are
usually not essential, except in times of illness, stress or for someone
challenged with a lifelong medical condition.
Essential amino acids are leucine,
isoleucine,
valine,
lysine,
threonine,
tryptophan,
methionine,
phenylalanine
and histidine.
Non-essential amino acids include alanine, asparagine, aspartic acid
and glutamic acid.
Conditional amino acids include arginine, cysteine, glutamine, glycine, proline, serine, and tyrosine.
Amino acids are found in animal sources such as meats,
milk, fish and eggs, as well as in plant sources such as whole grains, pulses,
legumes, soy, fruits, nuts and seeds. Vegetarians and vegans can get enough
essential amino acids by eating a variety of plant proteins.
Protein functions in body
Protein is a nutrient needed by the human body for growth
and maintenance. Aside from water, proteins are the most abundant kind of
molecules in the body. Protein can be found in all cells of the body and is the
major structural component of all cells in the body, especially muscle. This
also includes body organs, hair and skin. Proteins also are utilized in
membranes, such as glycoproteins. When broken down into amino
acids, they are used as precursors to nucleic acid,
co-enzymes, hormones, immune response, cellular repair and molecules essential
for life. Finally, protein is needed to form blood cells.
Proteins are very important molecules in our cells. They
are involved in virtually all cell functions. Each protein within the body has
a specific function. Some proteins are involved in structural support, while
others are involved in bodily movement, or in defense against germs. Proteins
vary in structure as well as function. They are constructed from a set of 20
amino acids and have distinct three-dimensional shapes. Below is a list of
several types of proteins and their functions.
Antibodies - are
specialized proteins involved in defending the body from antigens (foreign
invaders). They can travel through the blood stream and are utilized by the
immune system to identify and defend against bacteria, viruses, and other
foreign intruders. One way antibodies counteract antigens is by immobilizing
them so that they can be destroyed by white blood cells.
Contractile
Proteins - are responsible for movement. Examples include actin
and myosin. These proteins are involved in muscle contraction and movement.
Enzymes
- are proteins that facilitate biochemical reactions. They are often
referred to as catalysts because they speed up chemical reactions. Examples
include the enzymes lactase and pepsin. Lactase breaks down the sugar lactose
found in milk. Pepsin is a digestive enzyme that works in the stomach to break
down proteins in food.
Hormonal
Proteins - are messenger proteins which help to coordinate
certain bodily activities. Examples include insulin, oxytocin, and
somatotropin. Insulin regulates glucose metabolism by controlling the
blood-sugar concentration. Oxytocin stimulates contractions in females during
childbirth. Somatotropin is a growth hormone that stimulates protein production
in muscle cells.
Structural
Proteins - are fibrous and stringy and provide support. Examples
include keratin, collagen, and elastin. Keratins strengthen protective
coverings such as hair, quills, feathers, horns, and beaks. Collagens and elastin
provide support for connective tissues such as tendons and ligaments.
Storage
Proteins - store amino acids. Examples include ovalbumin and
casein. Ovalbumin is found in egg whites and casein is a milk-based protein.
Transport
Proteins - are carrier proteins which move molecules from one
place to another around the body. Examples include hemoglobin and cytochromes.
Hemoglobin transports oxygen through the blood. Cytochromes operate in the
electron transport chain as electron carrier proteins.
Protein function in
exercise
Proteins
are one of the key nutrients for success in terms of sports. Amino acids, the
building blocks of proteins, are used for building tissue, including muscle, as
well as repairing damaged tissues. Proteins usually only provide a small source
of fuel for the exercising muscles, being used as fuel typically only when
carbohydrates and lipid resources are low.
Sources
Animal sources of protein.
A wide range of foods are a source of protein. The best combination
of protein sources depends on the region of the world, access, cost, amino acid
types and nutrition balance, as well as acquired tastes. Some foods are high in
certain amino acids, but their digestibility and the anti-nutritional factors present in these
foods make them of limited value in human nutrition. Therefore, one must
consider digestibility and secondary nutrition profile such as calories, cholesterol,
vitamins and essential mineral density of the protein source. On a worldwide
basis, plant protein foods contribute over 60 percent of the per capita supply
of protein, on average. In
Meat, eggs and fish are sources of complete protein. Milk
and milk-derived foods are also good sources of protein.
Whole grains and cereals are another source of proteins.
However, these tend to be limiting in the amino acid lysine or threonine, which
are available in other vegetarian sources and meats. Examples of food staples
and cereal sources of protein, each with a concentration greater than 7
percent, are (in no particular order) buckwheat, oats, rye, millet, maize
(corn), rice, wheat, spaghetti, bulgar, sorghum, amaranth, and quinoa.
Vegetarian sources of proteins include legumes, nuts,
seeds and fruits. Legumes,
some of which are called pulses in certain parts of the world, have
higher concentrations of amino acids and are more complete sources of protein
than whole grains and cereals. Examples of vegetarian foods with protein
concentrations greater than 7 percent include soybeans, lentils, kidney beans,
white beans, mung beans, chickpeas, cowpeas, lima beans, pigeon peas, lupines,
wing beans, almonds, Brazil nuts, cashews, pecans, walnuts, cotton seeds,
pumpkin seeds, sesame seeds, and sunflower seeds.
Plant sources of protein.
Food staples that are poor sources of protein include
roots and tubers such as yams, cassava
and sweet potato.
Plantains, another major staple, are also a
poor source of essential amino acids. Fruits, while rich in other essential
nutrients, are another poor source of amino acids per
A good source of protein is often a
combination of various foods, because different foods are rich in different
amino acids. A good source of dietary protein meets two requirements:
·
The requirement for the nutritionally
indispensable amino acids (histidine, isoleucine, leucine, lysine, methionine,
phenylalanine, threonine, tryptophan, and valine) under all conditions and for
conditionally indispensable amino acids (cystine, tyrosine, taurine, glycine,
arginine, glutamine, proline) under specific physiological and pathological
conditions
·
The requirement for nonspecific nitrogen
for the synthesis of the nutritionally dispensable amino acids (aspartic acid,
asparagine, glutamic acid, alanine, serine) and other physiologically important
nitrogen-containing compounds such as nucleic acids, creatine, and porphyrins.
Healthy people eating a balanced diet
rarely need protein supplements. Except for a few amino acids, most are readily
available in human diet. The limiting amino acids are lysine, threonine,
tryptophan and sulfur-containing amino acids.
The table below presents the most important food groups
as protein sources, from a worldwide perspective. It also lists their
respective performance as source of the commonly limiting amino acids, in
milligrams of limiting amino acid per gram of total protein in the food source.
The green highlighted cells represent the protein source with highest density
of respective amino acid, while the yellow highlighted cells represent the
protein source with lowest density of respective amino acid. The table
reiterates the need for a balanced mix of foods to ensure adequate amino acid
source.
Protein milkshakes, made from protein powder (center) and milk (left), are
a common bodybuilding supplement.
Protein powders – such as casein, whey, egg,
rice and soy – are processed and manufactured
sources of protein. These protein powders may provide an additional source of
protein for bodybuilders. The type of protein is important in terms of its
influence on protein metabolic response and possibly on the muscle's exercise
performance. The different physical and/or chemical properties within the
various types of protein may affect the rate of protein digestion. As a result,
the amino acid availability and the accumulation of tissue protein is altered
because of the various protein metabolic responses.
Protein
quality
Different proteins have different levels
of biological availability (BA) to the human body. Many methods have been introduced
to measure protein utilization and retention rates in humans. They include biological
value, net protein utilization, and PDCAAS (Protein
Digestibility Corrected Amino Acids Score) which was developed by the FDA as an
improvement over the Protein efficiency ratio (PER) method.
These methods examine which proteins are most efficiently used by the body. The
PDCAAS rating is a fairly recent evaluation method; it was adopted by the US
Food and Drug Administration (FDA) and the Food and Agricultural Organization
of the United Nations/World Health Organization (FAO/WHO) in
1993 as "the preferred 'best'" method to determine protein quality.
These organizations have suggested that other methods for evaluating the
quality of protein are inferior.
Dietary
requirements
An education campaign launched by the United States Department of
Agriculture about 100 years ago, on cottage cheese as a lower-cost protein
substitute for meat.
Considerable debate has taken place regarding issues
surrounding protein intake requirements. The amount of protein required in a
person's diet is determined in large part by overall energy intake, the body's
need for nitrogen and essential amino acids, body weight and composition, rate
of growth in the individual, physical activity level, individual's energy and
carbohydrate intake, as well as the presence of illness or injury. Physical
activity and exertion as well as enhanced muscular mass increase the need for
protein. Requirements are also greater during childhood for growth and
development, during pregnancy or when breast-feeding in order to nourish a
baby, or when the body needs to recover from malnutrition or trauma or after an
operation.
If enough energy is not taken in through diet, as in the
process of starvation,
the body will use protein from the muscle mass to meet its energy needs,
leading to muscle wasting over time. If the individual does not consume
adequate protein in nutrition, then muscle will also waste as more vital
cellular processes (e.g. respiration enzymes, blood cells) recycle muscle
protein for their own requirements.
According to US & Canadian Dietary Reference Intake guidelines, women
aged 19–70 need to consume
Several studies have concluded that active people and
athletes may require elevated protein intake (compared to 0.8 g/kg) due to
increase in muscle mass and sweat losses, as well as need for body repair and
energy source. Suggested amounts vary between 1.6 g/kg and 1.8 g/kg, while a
proposed maximum daily protein intake would be approximately 25% of
energy requirements i.e. approximately 2 to 2.5 g/kg. However, many questions
still remain to be resolved.
The result of limited synthesis and normal rates
of protein degradation is that the balance of nitrogen intake and nitrogen
excretion is rapidly and significantly altered. Normal, healthy adults are
generally in nitrogen balance, with intake and excretion being very well
matched. Young growing children, adults recovering from major illness, and
pregnant women are often in positive nitrogen balance. Their intake of nitrogen
exceeds their loss as net protein synthesis proceeds. When more nitrogen is
excreted than is incorporated into the body, an individual is in negative
nitrogen balance. Insufficient quantities of even one essential amino acid is
adequate to turn an otherwise normal individual into one with a negative
nitrogen balance. The biological value of dietary proteins is related to the
extent to which they provide all the necessary amino acids. Proteins of animal
origin generally have a high biological value; plant proteins have a wide range
of values from almost none to quite high. In general, plant proteins are
deficient in lysine, methionine, and tryptophan and are much less concentrated
and less digestible than animal proteins. The absence of lysine in low-grade
cereal proteins, used as a dietary mainstay in many underdeveloped countries,
leads to an inability to synthesize protein (because of missing essential amino
acids) and ultimately to a syndrome known as kwashiorkor, common among children
in these countries.
Protein deficiency
A child in Africa suffering from kwashiorkor
– one of the three protein energy malnutrition ailments
afflicting over 10 million children in developing countries.
Protein deficiency and malnutrition can
lead to variety of ailments including mental retardation and kwashiorkor. Symptoms of
kwashiorkor include apathy, diarrhea, inactivity, failure to grow, flaky skin,
fatty liver, and edema of the belly and legs. This edema is explained by the
action of lipoxygenase on arachidonic acid to form leukotrienes and the normal
functioning of proteins in fluid balance and lipoprotein transport.
Although protein energy malnutrition is more common in
low-income countries, children from higher-income countries are also affected,
including children from large urban areas in low socioeconomic neighborhoods.
This may also occur in children with chronic diseases, and children who are
institutionalized or hospitalized for a different diagnosis. Risk factors
include a primary diagnosis of mental retardation, cystic fibrosis, malignancy,
cardiovascular disease, end stage renal disease, oncologic disease, genetic
disease, neurological disease, multiple diagnoses, or prolonged
hospitalization. In these conditions, the challenging nutritional management
may get overlooked and underestimated, resulting in an impairment of the
chances for recovery and the worsening of the situation.
Deficiency of protein leads to
following:
1. Shortage of protein leads to
retardation of growth and in extreme cases failure of growth. This is
manifested as marasmus and kwashiorkor among infants and children.
2. Protein deficiency affects the
intestinal mucosa and the gland that secret digestive enzymes. This results in
the failure to digest and absorb the food, consequently leading to diarrhea and
loss of fluid and electrolyte.
3. The normal structure and function of
liver is disturbed leading fat accumulation and fatty livers. Liver fails to
synthesis plasma albumin thus leading to Oedema.
4. Muscle wasting and anemia due to the
shortage of hemoglobin are common feature due to the deficiency of protein.
6. The amino acids presents in the
protein help in tissue synthesis during growth period e.g. infancy childhood
and adolescence. The body goes into negative N2 -balance due to the shortage of
protein in the diet. This results in muscle wastage
7. Proteins from important constituents
of hormones. How-ever the deficiency of proteins leads to no marked and
characteristic changes in the functioning of endocrine glands.
8. Proteins furnish 10-12per cent of
calories required daily. However the major part of proteins is essentially for
body-building purposes only.
Nitrogen balance
The nitrogen balance index (NBI) is used to evaluate the
amount of protein used by the body in comparison with the amount of protein
supplied from daily food intake. The body is in the state of nitrogen (or
protein) equilibrium when the intake and usage of protein is equal. The body
has a positive nitrogen balance
when the intake of protein is greater than that expended by the body. In this
case, the body can build and develop new tissue. Since the body does not store
protein, the overconsumption of protein can result in the excess amount to be
converted into fat and stored as adipose tissue.
Blood urea nitrogen can be used in estimating
nitrogen balance.
A positive
value is often found during periods of growth, tissue repair or pregnancy.

This means that the intake of nitrogen into the body
is greater than the loss of nitrogen from the body, so there is an increase in
the total body pool of protein.
The
body has a negative nitrogen balance when the intake of protein is less
than that expended by the body. In this case, protein intake is less than
required, and the body cannot maintain or build new tissues.
A negative
nitrogen balance represents a state of protein deficiency, in which the
body is breaking down tissues faster than they are being replaced. The
ingestion of insufficient amounts of protein, or food with poor protein
quality, can result in serious medical conditions in which an individual's
overall health is compromised. The immune system is severely affected;
the amount of blood plasma decreases, leading to medical conditions such
as anemia or edema; and the body becomes vulnerable to infectious
diseases and other serious conditions. Protein malnutrition in infants is
called kwashiorkor, and it poses a major health problem in developing
countries, such as Africa, Central and South America, and certain parts of
A
negative value can be associated with burns, fevers, wasting diseases and other
serious injuries and during periods of fasting. This means that the amount of
nitrogen excreted from the body is greater than the amount of nitrogen ingested.

It can be used in the evaluation of malnutrition.
The difference
between the total
nitrogen
intake by an organism
and its total nitrogen loss. A normal,
healthy
adult
has a zero
nitrogen balance.
Essential and nonessential
amino acids.
THE TWENTY AMINO ACIDS
|
Name |
Abbreviation |
Linear structure formula (atom composition and bonding) |
|
SOURCE:
Institute for Chemistry |
||
|
Alanine |
ala |
CH3-CH(NH2)-COOH
|
|
Arginine |
arg |
HN=C(NH2)- |
|
Asparagine |
asn |
H2N-CO-CH2-CH(NH2)-COOH |
|
Aspartic acid |
asp |
HOOC-CH2-CH(NH2)-COOH |
|
Cysteine |
cys |
HS-CH2-CH(NH2)-COOH |
|
Glutamine |
gln |
H2N- |
|
Glutamic acid |
glu |
HOOC-(CH2)2-CH(NH2)-COOH |
|
Glycine |
gly |
NH2-CH2-COOH
|
|
Histidine |
his |
NH-CH=N-CH=C-CH2-CH(NH2)-COOH |____________| |
|
Isoleucine |
ile |
CH3-CH2-CH(CH3)-CH(NH2)-COOH |
|
Leucine |
leu |
(CH3)2-CH-CH2-CH(NH2)-COOH |
|
Lysine |
lys |
H2N-(CH2)4-CH(NH2)-COOH |
|
Methionine |
met |
CH3-S-(CH2)2-CH(NH2)-COOH |
|
Phenylalanine |
phe |
Ph-CH2-CH(NH2)-COOH |
|
Proline |
pro |
NH-(CH2)3-CH-COOH
|__________| |
|
Serine |
ser |
HO-CH2-CH(NH2)-COOH |
|
Threonine |
thr |
CH3-CH(OH)-CH(NH2)-COOH |
|
Tryptophan |
trp |
Ph-NH-CH=C-CH2-CH(NH2)-COOH |_________| |
|
Tyrosine |
tyr |
HO-Ph-CH2-CH(NH2)-COOH |
|
Valine |
val |
(CH3)2-CH-CH(NH2)-COOH |
consist of a sufficient and
balanced supply of both essential and
nonessential amino acids in order to ensure high levels of protein
production.
Essential
vs. Nonessential Amino Acids
|
Nonessential |
Essential |
|
Alanine |
Arginine* |
|
Asparagine |
Histidine |
|
Aspartate |
Isoleucine |
|
Cysteine |
Leucine |
|
Glutamate |
Lysine |
|
Glutamine |
Methionine* |
|
Glycine |
Phenylalanine* |
|
Proline |
Threonine |
|
Serine |
Tryptophan |
|
Tyrosine |
Valine |
|
The amino
acids arginine, methionine and phenylalanine are considered essential for
reasons not directly related to lack of synthesis. Arginine is synthesized by
mammalian cells but at a rate that is insufficient to meet the growth needs
of the body and the majority that is synthesized is cleaved to form urea.
Methionine is required in large amounts to produce cysteine if the latter
amino acid is not adequately supplied in the diet. Similarly, phenyalanine is
needed in large amounts to form tyrosine if the latter is not adequately
supplied in the diet. |
|
The quality of protein depends on the level at which it provides the
nutritional amounts of essential amino acids needed for overall body health,
maintenance, and growth. Animal proteins, such as eggs, cheese, milk, meat, and
fish, are considered high-quality, or complete, proteins because
they provide sufficient amounts of the essential amino acids. Plant proteins,
such as grain, corn, nuts, vegetables and fruits, are lower-quality, or incomplete,
proteins because many plant proteins lack one or more of the essential
amino acids, or because they lack a proper balance of amino acids. Incomplete
proteins can, however, be combined to provide all the essential amino acids,
though combinations of incomplete proteins must be consumed at the same time,
or within a short period of time (within four hours), to obtain the maximum
nutritive value from the amino acids. Such combination diets generally yield a
high-quality protein meal, providing sufficient amounts and proper balance of
the essential amino acids needed by the body to function.
Digestion of proteins in stomach and small intestine.


Digestion in the stomach
Chemical digestion begins in the stomach. The stomach is a large,
hollow, pouched-shaped muscular organ. Food in the stomach is broken down by
the action of gastric juice, which
contains hydrochloric acid and pepsin (an enzyme that digests protein). The
stomach begins its production of gastric juice while food is still in the
mouth. Nerves from the cheeks and tongue are stimulated and send messages to
the brain. The brain in turn sends messages to nerves in the stomach wall,
stimulating the secretion of gastric juice before the arrival of food. The
second signal for gastric juice production occurs when food arrives in the
stomach and touches the lining.
Gastric juice is secreted from the linings of the stomach walls, along
with mucus that helps to protect the stomach lining from the action of the
acid. Three layers of powerful stomach muscles churn food into a thick liquid
called chyme (pronounced KIME). From time to time, chyme is passed through the
pyloric sphincter, the opening between the stomach and the small intestine.


Protein digestion begins when the food reaches the stomach and
stimulates the release of hydrochloric acid (HCl) by the parietal cells located
in the gastric mucosa of the GI (gastrointestinal) tract.
Hydrochloric acid provides for a very acidic environment, which helps
the protein digestion process in two ways:

(1) through
an acid-catalyzed hydrolysis reaction of breaking peptide bonds (the
chemical process of breaking peptide bonds is referred to as a hydrolysis
reaction because water is used to break the bonds); and (2) through conversion
of the gastric enzyme pepsinogen (an inactive precursor) to pepsin (the
active form). Pepsinogen is stored and secreted by the "chief cells"
that line the stomach wall. Once converted into the active form, pepsin attacks
the peptide bonds that link amino acids together, breaking the long polypeptide
chain into shorter segments of amino acids known as dipeptides and tripeptides.
These protein fragments are then further
broken down in the duodenum of the small intestines.

(2) The
brush border enzymes, which work on the surface of epithelial cells
of the small intestines, hydrolyze the protein fragments into amino
acids.

Digestion and absorption in the small intestine
The small
intestine is a long, narrow tube running from the stomach to the large
intestine. The small intestine is greatly coiled and twisted. Its full length
is about

The
duodenum is about
The amino
acids travel through the hepatic portal vein to the liver, where the nutrients
are processed into glucose or fat (or released into the
bloodstream). The tissues in the body take up the amino acids rapidly for
glucose production, growth and maintenance, and other vital cellular
functioning. For the most part, the body does not store protein, as the
metabolism of amino acids occurs within a few hours.
Amino acids
are metabolized in the liver into useful forms that are used as building blocks
of protein in tissues. The body may utilize the amino acids for either anabolic
or catabolic reactions. Anabolism refers to the chemical process through
which digested and absorbed products are used to effectively build or repair
bodily tissues, or to restore vital substances broken down through metabolism. Catabolism,
on the other hand, is the process that results in the release of energy through
the breakdown of nutrients, stored materials, and cellular substances. Anabolic
and catabolic reactions work hand-in-hand, and the energy produced in catabolic
processes is used to fuel essential anabolic processes. The vital biochemical
reaction of glycolysis (in which glucose is oxidized to produce carbon
dioxide, water, and cellular energy) in the form of adenosine triphosphate, or
ATP, is a prime example of a catabolic reaction. The energy released, as ATP,
from such a reaction is used to fuel important anabolic processes, such as
protein synthesis.
The chyme first enters duodenum, which
is a part of small intestine. It is a C-shaped structure about
Trypsin is a serine
protease found in the digestive
system of many vertebrates, where it hydrolyses
proteins.
Trypsin is produced in the pancreas as the inactive proenzyme
trypsinogen.
Trypsin cleaves peptide
chains mainly at the carboxyl side of the amino acids
lysine
or arginine,
except when either is followed by proline. It is used for numerous biotechnological
processes. The process is commonly referred to as trypsin proteolysis
or trypsinisation,
and proteins that have been digested/treated with trypsin are said to have been
trypsinized.
Function
In the duodenum,
trypsin catalyzes
the hydrolysis
of peptide bonds,
breaking down proteins into smaller peptides. The peptide products are then
further hydrolyzed into amino acids via other
proteases, rendering them available for absorption into the blood
stream. Tryptic digestion is a necessary step in protein absorption as proteins
are generally too large to be absorbed through the lining of the small
intestine.
Trypsin is produced in the pancreas,
in the form of the inactive zymogen trypsinogen. When the pancreas is stimulated by
cholecystokinin,
it is then secreted into the first part of the small intestine (the duodenum)
via the pancreatic duct. Once in the small intestine,
the enzyme enteropeptidase activates it into trypsin by proteolytic cleavage. Auto catalysis does
not happen with trypsin since trypsinogen is a poor substrate for trypsin. This
activation mechanism is common for most serine proteases, and serves to prevent
autodegradation of the
pancreas.
Mechanism
The enzymatic mechanism is similar to
that of other serine proteases. These enzymes contain a catalytic
triad consisting of histidine-57, aspartate-102,
and serine-195.
These three
residues form a charge relay that serves to make the active site
serine
nucleophilic. This is achieved by modifying the electrostatic environment of
the serine. The enzymatic reaction that trypsins catalyze is thermodynamically
favorable but requires significant activation
energy (it is "kinetically
unfavorable"). In addition, trypsin contains an "oxyanion hole"
formed by the backbone amide hydrogen atoms of Gly-193 and Ser-195,
which serves to stabilize the developing negative charge on the carbonyl oxygen
atom of the cleaved amides.
The aspartate
residue (Asp 189) located in the catalytic pocket (S1) of trypsins is
responsible for attracting and stabilizing positively charged lysine and/or arginine,
and is, thus, responsible for the specificity of the enzyme. This means that
trypsin predominantly cleaves proteins at the carboxyl side (or "C-terminal
side") of the amino acids lysine and arginine
except when either is bound to a C-terminal proline.,
although large-scale mass spectrometry data suggest cleavage occurs even with
proline. Trypsins are considered endopeptidases,
i.e., the cleavage occurs within the polypeptide
chain rather than at the terminal amino acids located at the ends of polypeptides.
Properties
Trypsins has an optimal operating pH of about 7.5-8.5 and
optimal operating temperature of about
The activity of trypsins is not affected by the inhibitor
tosyl phenylalanyl chloromethyl ketone, TPCK, which deactivates chymotrypsin.
This is important because, in some applications, like mass
spectrometry, the specificity of cleavage is important.
Trypsins should be stored at very cold temperatures
(between −20°C and −80°C) to prevent autolysis,
which may also be impeded by storage of trypsins at pH 3 or by using trypsin
modified by reductive methylation. When
the pH is adjusted back to pH 8, activity returns.
Clinical significance
Activation of trypsin from proteolytic
cleavage of trypsinogen in the pancreas can lead to a series of events that
cause pancreatic self-digestion, resulting in pancreatitis.
One consequence of the autosomal recessive disease cystic
fibrosis is a deficiency in transport of trypsin and other digestive
enzymes from the pancreas. This leads to the disorder termed meconium
ileus. This disorder involves intestinal obstruction (ileus) due to overly thick
meconium,
which is normally broken down by trypsins and other proteases, then passed in
faeces.
Trypsin is available in high quantity in
pancreases, and can be purified rather easily. Hence it has been used widely in
various biotechnological processes.
In a tissue
culture lab, trypsins are used to re-suspend cells adherent to the
cell culture dish wall during the process of harvesting cells. Some cell types
have a tendency to "stick" - or adhere - to the sides and bottom of a
dish when cultivated in vitro. Trypsin is used to cleave proteins bonding the
cultured cells to the dish, so that the cells can be suspended in fresh
solution and transferred to fresh dishes.
Trypsin can also be used to dissociate
dissected cells (for example, prior to cell fixing and sorting).
Trypsins can be used to break down
casein in breast milk. If trypsin is added to a solution of milk powder, the
breakdown of casein will cause the milk to become translucent.
The rate of reaction can be measured by using the amount of time it takes for
the milk to turn translucent.
Trypsin is commonly used in biological
research during proteomics experiments to digest proteins into peptides for
mass spectrometry analysis, e.g. in-gel
digestion. Trypsin is particularly suited for this, since it has a
very well defined specificity, as it hydrolyzes only the peptide bonds in which
the carbonyl group is contributed either by an Arg or
Trypsin can also be used to dissolve blood
clots in its microbial form and treat inflammation in its pancreatic form.
Rotting of proteins in a large
intestine, products.
The large
intestine performs the following functions:
- reabsorbs water and maintains the
fluid balance of the body
·
- absorbs certain vitamins
·
- processes undigested material (fibre)
·
- stores waste before it is eliminated.
The
large intestine is wider and heavier than the small intestine. However, it is
much shorter—only about

The
large intestine removes water from the waste products of digestion and returns
some of it to the bloodstream. Fecal matter contains undigested food, bacteria,
and cells from the walls of the digestive tract. Millions of bacteria in the
large intestine help to produce certain B vitamins and vitamin K. These
vitamins are absorbed into the bloodstream along with the water.
The caecum is
the first part of the large intestine. Shaped like a small pouch and located in
the right lower abdomen, it is the connection between the small intestine and
the colon.
The caecum accepts and stores processed
material from the small intestine and moves it towards the colon. As the
processed food approaches the end of the small intestine, a valve separating
the small and large intestines opens, the caecum expands and the material
enters. At this stage, the mixture normally contains:
·
undigested food (fibre)
·
a little bit of water
·
some vitamins
·
some minerals or salts
The metabolism of amino acids can be understood from the dynamic
catabolic and anabolic processes. In the process referred to as deamination,
the nitrogen-containing amino group (NH2) is cleaved from the amino
acid unit. In this reaction, which requires vitamin B6 as a cofactor, the amino
group is transferred to an acceptor keto-acid, which can form a new
amino acid. Through this process, the body is able to make the nonessential
amino acids not provided by one's diet. The keto-acid intermediate can also be
used to synthesize glucose to ultimately yield energy for the body, and the
cleaved nitrogen-containing group is transformed into urea, a waste product,
and excreted as urine.
Proteins are vital to basic cellular
and body functions, including cellular regeneration and repair, tissue
maintenance and regulation, hormone and enzyme production, fluid
balance, and the provision of energy.
THE KINDS OF
GASTRIC JUICE ACIDITY.
Gastric acid is a digestive
fluid, formed in the stomach. It has a pH of 1.5 to 3.5 and is composed of hydrochloric
acid (HCl) (around 0.5%, or 5000 parts per
million) as high as 0.1 N, and large quantities of potassium chloride (KCl) and sodium
chloride (NaCl). The acid plays a key role in digestion of proteins,
by activating digestive enzymes, and making ingested proteins
unravel so that digestive enzymes break down the long chains of amino acids.
Gastric acid is produced by cells lining the stomach,
which are coupled to systems to increase acid production when needed. Other
cells in the stomach produce bicarbonate, a base, to buffer
the fluid, ensuring that it does not become too acidic. These cells also
produce mucus,
which forms a viscous physical barrier to prevent
gastric acid from damaging the stomach. Cells in the beginning of the
small intestine, or duodenum, further produce large amounts of bicarbonate to
completely neutralize any gastric acid that passes further down into the
digestive tract.
Gastric acid is produced by parietal
cells (also called oxyntic cells) in the stomach. Its secretion is a
complex and relatively energetically expensive process. Parietal cells contain
an extensive secretory network (called canaliculi) from which the gastric acid is
secreted into the lumen of the stomach. These cells are part of epithelial
fundic glands
in the gastric mucosa. The pH of gastric acid is 1.35
to 3.5 [2]
in the human stomach lumen, the acidity being maintained by the proton pump
H+/K+ ATPase. The
parietal cell releases bicarbonate into the blood stream in the
process, which causes a temporary rise of pH in the blood, known as alkaline tide.
The resulting highly acidic environment
in the stomach lumen causes proteins from food to lose their characteristic folded
structure (or denature). This exposes the protein's peptide bonds.
The chief cells
of the stomach secrete enzymes for protein breakdown (inactive pepsinogen
and rennin).
Hydrochloric acid activates pepsinogen into the
enzyme
pepsin,
which then helps digestion by breaking the bonds linking amino acids,
a process known as proteolysis. In addition, many microorganisms
have their growth inhibited by such an acidic environment, which is helpful to
prevent infection.
Secretion
There are three phases in the secretion of gastric acid:
1.
The basal phase: A small amount of acid
is always being secreted into the stomach. The three following phases increase
the secretion rate in order to digest a meal.
2.
The cephalic phase: Thirty percent of
the total gastric acid secretions to be produced is stimulated by anticipation
of eating and the smell or taste of food. This signalling occurs from higher
centres in the brain through the Vagus Nerve.
It activates parietal cells to release acid and ECL cells
to release histamine.
The Vagus nerve also releases Gastrin Releasing Peptide onto G cells.
Finally, it also inhibits somatostatin release from D cells.
3.
The gastric phase: About fifty percent of
the total acid for a meal is secreted in this phase. Acid secretion is
stimulated by distension of the stomach and by amino acids
present in the food.
4.
The intestinal phase: The remaining 10%
of acid is secreted when chyme enters the small intestine, and is stimulated by small
intestine distension and by amino acids. The duodenal cells release entero-oxyntin
which acts on parietal cells without affecting gastrin.
There is also a small continuous basal secretion of
gastric acid between meals of usually less than 10 mEq/hour.
Regulation of secretion
Diagram depicting the major determinants of gastric acid secretion, with inclusion
of drug targets for peptic ulcer disease (PUD) and gastroesophageal reflux
disease (GERD).
Gastric acid production is regulated by
both the autonomic nervous system and several hormones.
The parasympathetic nervous system,
via the vagus nerve,
and the hormone gastrin
stimulate the parietal cell to produce gastric acid, both directly acting on
parietal cells and indirectly, through the stimulation of the secretion of the
hormone histamine
from enterochromaffine-like cells (ECL). Vasoactive intestinal peptide, cholecystokinin,
and secretin
all inhibit production.
The production of gastric acid in the
stomach is tightly regulated by positive regulators and negative
feedback mechanisms. Four types of cells are involved in this
process: parietal cells, G cells, D cells
and enterochromaffine-like cells. Besides this, the endings of the vagus nerve
(CN X) and the intramural nervous plexus in the digestive tract influence the
secretion significantly.
Nerve endings in the stomach secrete two
stimulatory neurotransmitters: acetylcholine
and gastrin-releasing peptide. Their action is
both direct on parietal cells and mediated through the secretion of gastrin
from G cells and histamine from enterochromaffine-like cells. Gastrin acts on
parietal cells directly and indirectly too, by stimulating the release of
histamine.
The release of histamine is the most
important positive regulation mechanism of the secretion of gastric acid in the
stomach. Its release is stimulated by gastrin and acetylcholine and inhibited
by somatostatin.
Neutralization
In the duodenum, gastric acid is neutralized by sodium bicarbonate. This also blocks gastric
enzymes that have their optima in the acid range of pH. The secretion of
sodium bicarbonate from the pancreas is stimulated by secretin.
This polypeptide
hormone gets activated and secreted from so-called S cells
in the mucosa of the duodenum and jejunum when the pH in duodenum falls below 4.5 to 5.0. The
neutralization is described by the equation:
HCl + NaHCO3 → NaCl + H2CO3
The carbonic acid rapidly equilibrates with carbon
dioxide and water through catalysis by carbonic anhydrase
enzymes bound to the gut epithelial lining,[6]
leading to a net release of carbon dioxide gas within the lumen associated with
neutralisation. In the absorptive upper intestine, such as the duodenum, both
the dissolved carbon dioxide and carbonic acid will tend to equilibrate with
the blood, leading to most of the gas produced on neutralisation being exhaled
through the lungs.
Role in disease
In hypochlorhydria and achlorhydria,
there is low or no gastric acid in the stomach, potentially leading to problems
as the disinfectant
properties of the gastric lumen are decreased. In such conditions, there is
greater risk of infections of the digestive
tract (such as infection with Vibrio or Helicobacter
bacteria).
In Zollinger–Ellison syndrome and hypercalcemia,
there are increased gastrin levels, leading to excess gastric acid production,
which can cause gastric ulcers.
In diseases featuring excess vomiting, patients develop hypochloremic
metabolic alkalosis (decreased blood acidity by
H+
and chlorine
depletion).

Your stomach is where the
food you eat is broken down into smaller pieces. This action is called
digestion.
Stomach (gastric) juice is the term used to describe the chemicals that
break down food in the stomach. These include hydrochloric acid and an enzyme
called pepsin. Gastric juice is sometimes referred to as stomach acid, although
not all of the substances in gastric juice are acidic.

Hypoaciditas,
hyperaciditas, anaciditas, hypochlorhydria, hyperchlorhydria, achlorhydria.
HYPOCHLORHYDRIA
(Obvious) is the lack of adequate production of Hydrochloric Acid (HCL) by the
Stomach Parietal Cells.

Many
people, in the process of aging, develop various stages of Hypochlorhydria;
however, it is not confined to this aging group. Many young people also
develop this problem. Bear in mind that the presence of HCL in the
Stomach generally inhibits (slows down or stops) the reflex of “rapid-dumping”
of foods out of the Stomach, rendering the critical First Stage of Digestion
partially or totally incomplete. Also, HCL performs a natural sterilization of
the foods that we swallow. This is quite important, because nothing that we eat
is sterile. In the pre-digestion phase of the Stomach, HCL, Pepsin, certain
Enzymes, plus the Intrinsic Factor, which is essential for the absorption of
Vitamin B-12, play key rolls in the conversion processes of Proteins to Amino
Acids and Starches to Sugars that can be utilized by our bodies (in conjunction
with the Duodenal, 2nd Phase of Digestion).
ACHLORHYDRIA
ACHLORHYDRIA
is the total absence of HCL production in the stomach.
Patients
with Achlorhydria May Have a form of Pernicious Anemia. This will also
show in a routine Blood test. When the Anemia is corrected the Stomachs
Parietal Function will generally return to
HYPERCHLORHYDRIA

HYPERCHLORHYDRIA,
The above Graph indicates the excess production of
Hydrochloric Acid (HCL). This condition may cause Delayed, or Marked-delayed,
emptying time of the Stomach's contents. In many cases, Patients with Delayed
and Marked-delayed emptying, will retain food in their Stomachs for 6 to 24
hours, or much longer in many cases.
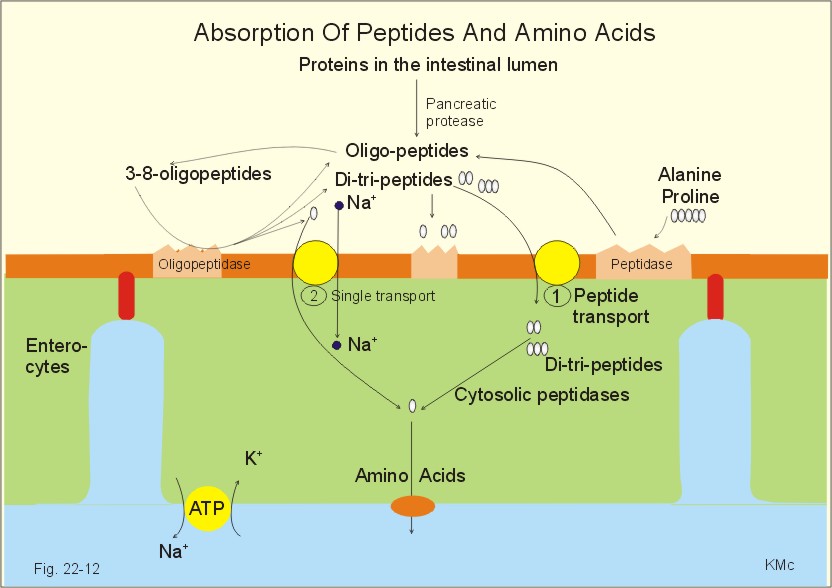
Absorption of Amino Acids and Peptides
Dietary proteins are, with very few exceptions, not absorbed. Rather, they
must be digested into amino acids or di- and tripeptides first. In previous
sections, we've seen two sources secrete proteolytic enzymes into the lumen of
the digestive tube:
·
the stomach
secretes pepsinogen, which is converted to the active protease
pepsin by the action of acid.
·
the pancreas
secretes a group of potent proteases, chief among them trypsin,
chymotrypsin and carboxypeptidases.
Through the action of these gastric and pancreatic proteases, dietary
proteins are hydrolyzed within the lumen of the small intestine predominantly
into medium and small peptides (oligopeptides).
The brush border of the small intestine is equipped with a family of
peptidases. Like lactase and maltase, these peptidases are integral membrane
proteins rather than soluble enzymes. They function to further the hydrolysis
of lumenal peptides, converting them to free amino acids and very small
peptides. These endproducts of digestion, formed on the surface of the
enterocyte, are ready for absorption.
Absorption of Amino Acids
The mechanism by which amino acids are absorbed is conceptually
identical to that of monosaccharides. The lumenal plasma membrane of
the absorptive cell bears at least four sodium-dependent amino acid
transporters - one each for acidic, basic, neutral and amino acids. These
transporters bind amino acids only after binding sodium. The fully loaded
transporter then undergoes a conformational change that dumps sodium and the
amino acid into the cytoplasm, followed by its reorientation back to the
original form.
Thus, absorption of amino acids is also absolutely dependent
on the electrochemical gradient of sodium across the epithelium. Further,
absorption of amino acids, like that of monosaccharides, contributes to
generating the osmotic gradient that drives water absorption.
The basolateral membrane of the enterocyte contains
additional transporters which export amino acids from the cell into blood.
These are not dependent on sodium gradients.
Absorption of Peptides
There is virtually no absorption of peptides longer than four amino acids.
However, there is abundant absorption of di- and tripeptides in the small
intestine. These small peptides are absorbed into the small intestinal
epithelial cell by cotransport with H+ ions via a transporter called
PepT1.
Once inside the enterocyte, the vast bulk of absorbed di- and tripeptides
are digested into amino acids by cytoplasmic peptidases and exported from the
cell into blood. Only a very small number of these small peptides enter blood
intact.
Absorption of Intact Proteins
As emphasized, absorption of intact proteins occurs only in a few
circumstances. In the first place, very few proteins get through the gauntlet
of soluble and membrane-bound proteases intact. Second, "normal"
enterocytes do not have transporters to carry proteins across the plasma membrane
and they certainly cannot permeate tight junctions.
One important exception to these general statements is that for a very few
days after birth, neonates have the ability to absorb intact proteins. This
ability, which is rapidly lost, is of immense importance because it allows the
newborn animal to acquire passive immunity by absorbing immunoglobulins in
colostral milk.
In constrast to humans and rodents, there is no significant transfer of
antibodies across the placenta
in many animals (cattle, sheep, horses and pigs to name a few), and the young
are born without circulating antibodies. If fed colostrum during the first day or
so after birth, they absorb large quantities of immunoglobulins and acquire a
temporary immune system that provides protection until they generate their own
immune responses.
The small intestine rapidly loses the capacity to absorb intact proteins -
a process called closure - and consequently, animals that do not receive
colostrum within the first few days after birth will likely die due to
opportunistic infections.
Intestinal barrier function regulates
transport and host defense mechanisms at the mucosal interface with the outside
world. Transcellular and paracellular fluxes are tightly controlled by membrane
pumps, ion channels and tight junctions, adapting permeability to physiological
needs. Permeability greatly defines the absorbance of compounds in the
intestine. In case of high permeability compounds it is unlikely that the
absorption will be modified by transporters, yet the test compound might be
involved in transporter mediated drug-drug interactions. The absorption
of medium and low permeability compounds can be affected by membrane
transporters located at the endothelial cells of the intestinal barrier.
In
the intestine transporters are localized in the brush border membrane and on
the basolateral side of intestinal cells. Four major ABC efflux transporters
have been shown to localize at the apical/luminal membrane of enterocytes, and
thus are thought to form a barrier to intestinal absorption of substrate drugs.


General pathways of amino acids transformation
Deamination of amino acids, it kinds. The role of
vitamins in deamination of amino acids.
Oxidative
Deamination Reaction
Deamination is also an oxidative reaction
that occurs under aerobic conditions in all tissues but especially the liver.
During oxidative deamination, an amino acid is converted into the
corresponding keto acid by the removal of the amine functional group as ammonia
and the amine functional group is replaced by the ketone group. The ammonia
eventually goes into the urea cycle.
Oxidative deamination occurs primarily on glutamic
acid because glutamic acid was the end product of many transamination
reactions.

The glutamate dehydrogenase is
allosterically controlled by ATP and ADP. ATP acts as an inhibitor whereas ADP
is an activator.
Central Role for Glutamic Acid:

Apparently most amino acids may be deaminated but this is a significant
reaction only for glutamic acid. If this is true, then how are the other amino
acids deaminated? The answer is that a combination of transamination and
deamination of glutamic acid occurs which is a recycling type of reaction for
glutamic acid. The original amino acid loses its amine group in the process. The general reaction sequence is
shown on the left.

Synthesis of New Amino Acids:
The same reaction works in reverse for the synthesis
of amino acids. In this situation alpha-ketoglutaric acid first uses
transamination of a different amino acid to make glutamic acid, which then
reacts with a keto acid to make a new amino acid. In effect, the
interconversion of alpha-ketoglutaric acid and glutamic acid lies at the very
heart of nitrogen metabolism. These molecules serve as the "collection and
receiving agent" for nitrogen. The subsequent fate of the amino group is
in new amino acids, any nitrogen bases, or any other nitrogen containing compounds.

Transamination of amino acids,
mechanism, role of enzymes and coenzymes.
Transamination
as the name implies, refers to the transfer of an amine group from one molecule
to another. This reaction is catalyzed by a family of enzymes called
transaminases. Actually, the transamination reaction results in the exchange of
an amine group on one acid with a ketone group on another acid. It is analogous
to a double replacement reaction.

Pyridoxine is an example of a vitamin which
is required for the synthesis of a coenzyme. Other examples which you have met
in this course are listed in the table below.
|
vitamin |
cofactor |
example enzymes |
|
pyridoxine (vit b6) |
pyridoxal phosphate |
transaminases |
|
niacin (nicotinamide) |
NAD+/ NADP+ |
malate dehydrogenase |
|
riboflavin (vit b2) |
FAD |
succinate dehydrogenase |
|
thiamine (vit b1) |
TPP |
pyruvate dehydrogenase |
|
pantothenate |
Coenzyme A |
fatty acid metabolism |
The most
usual and major keto acid involved with transamination reactions is
alpha-ketoglutaric acid, an intermediate in the citric acid cycle. A specific
example is the transamination of alanine to make pyruvic acid and glutamic
acid.
Other amino
acids which can be converted after several steps through transamination into
pyruvic acid include serine, cysteine, and glycine.

Other Transamination Reactions:
Aspartic acid can be converted into
oxaloacetic acid, another intermediate of the citric acid cycle. Other amino
acids such as glutamine, histidine, arginine, and proline are first converted
into glutamic acid.
Glutamine and asparagine are
converted into glutamic acid and aspartic acid by a simple hydrolysis of the
amide group.
All of the amino acids can be
converted through a variety of reactions and transamination into a keto acid
which is a part of or feeds into the citric acid cycle. The interrelationships
of amino acids with the citric acid cycle are illustrated in the graphic on the
left.

Amino Acids in Overall Metabolism:
Once the keto acids have been formed
from the appropriate amino acids by transamination, they may be used for
several purposes. The most obvious is the complete metabolism into carbon
dioxide and water by the citric acid cycle.
However, if there are excess proteins
in the diet those amino acids converted into pyruvic acid and acetyl CoA can be
converted into lipids by the lipogenesis process. If carbohydrates are lacking
in the diet or if glucose cannot get into the cells (as in diabetes), then
those amino acids converted into pyruvic acid and oxaloacetic acids can be
converted into glucose or glycogen.
The hormones cortisone and cortisol
from the adrenal cortex stimulate the synthesis of glucose from amino acids in
the liver and also function as antagonists to insulin

Synthesis of New Amino Acids:
In addition to the catabolic function
of transamination reactions, these reactions can also be used to synthesize
amino acids needed or not present in the diet. An amino acid may be synthesized
if there is an available "root" ketoacid with a synthetic connection
to the final amino acid. Since an appropriate "root" keto acid does
not exist for eight amino acids, (lys, leu, ile, met, thr, try, val, phe), they
are essential and must be included in the diet because they cannot be
synthesized.
Glutamic acid usually serves as the
source of the amine group in the transamination synthesis of new amino acids.
The reverse of the reactions mentioned earlier are the most obvious methods for
producing the amino acids alanine and aspartic acid.
Several nonessential amino acids are
made by processes other than transamination. Cysteine is made from methionine,
and serine and glycine are synthesized from phosphoglyceric acid - an
intermediate of glycolysis
The correlation between
transamination and deamination.
Pathways
Decarboxylisation of
amino acids, role of enzymes and co-enzymes.
Decarboxylation is any chemical
reaction in which a carboxyl group (-COOH) is split off from a compound as carbon
dioxide (CO2).
Common biosynthetic
decarboxylations of amino acids to amines are:
- tryptophan
to tryptamine
- phenylalanine
to phenylethylamine
- tyrosine
to tyramine
- histidine
to histamine
- serine
to ethanolamine
- glutamic
acid to GABA
- lysine
to cadaverine
- arginine
to agmatine
- ornithine
to putrescine
- 5-HTP
to serotonin
- L-DOPA
to dopamine
Other decarboxylation reactions from the citric acid
cycle include:
to succinyl-CoA.
Enzymes that catalyze
decarboxylations are called decarboxylases or, more formally, carboxy-lyases
. Carboxy-lyases, also known as decarboxylases, are carbon-carbon lyases that add
or remove a carboxyl
group from organic compounds. These enzymes catalyze
the decarboxylation of amino acids, beta-keto acids and alpha-keto acids
Heating or pyrolysis of
Δ9-Tetrahydrocannabinolic acid yields the psychoactive compound

Main bioactive
amines, their source and role in organism.
|
|
Biogenic amines are basic
nitrogenous low molecular weight compounds with biological activity that may be
formed or catabolised during the normal metabolism of animals, plants and
micro-organisms. Biogenic amines are derived mainly from amino acids through
substrate-specific decarboxylase enzymes. Amines may be formed by yeasts during
the alcoholic fermentation (mostly putrazine); by lactic acid bacteria (LAB)
during malolactic fermentation (MLF. Biogenic amines can also be present in the
must, just as putrescine in grapes is associated with potassium deficiencies in
the soil. The main biogenic amines are histamine, tyramine, putrescine,
cadaverine and phenylethylamine.
Microorganisms decarboxylise amino acids in order to provide the cell
with energy and to protect the cell against acidic environments by increasing
the pH.
Biogenic amines are important because they
contain a health risk for sensitive individuals. Symptoms include nausea,
respiratorial discomfort, hot flushes, cold sweat, palpitations, headaches, red
rash, high or low blood pressure. Alcohol and acetaldehyde have been found to
increase the sensitivity to biogenic amines.

Mast cells release
histamine when an allergen is encountered.
The histamine response
can produce sneezing, itching, hives and watery eyes.

Allergies.
Some antigens, termed allergens trigger
the release of IgE antibodies from specialized B cells. The antibodies
are inserted into the membrane of leucocytes called mast cells. If the
allergen is again presented to an activated mast cell, it binds to the cell
surface of the mast cell. This triggers the cell to release histamine and leucotrines that cause
allergic reactions

Serotonin
5-Hydroxytryptophan is decarboxylated to serotonin
(5-hydroxytryptamine or 5-HT) by the enzyme aromatic-L-amino-acid decarboxylase
with the help of vitamin B6. This reaction occurs both in nervous tissue and in
the liver. 5-HTP crosses the blood–brain barrier, while 5-HT does not. Excess
5-HTP, especially when administered with Vitamin B6, is thought to be
metabolized and excreted.
In animals including humans, serotonin is synthesized from the amino acid
L-tryptophan by a short metabolic pathway consisting of two enzymes: tryptophan
hydroxylase (TPH) and amino acid decarboxylase (DDC). The TPH-mediated reaction
is the rate-limiting step in the pathway. TPH has been shown to exist in two
forms: TPH1, found in several tissues, and TPH2, which is a brain-specific
isoform.
Serotonin taken orally does not pass into the serotonergic pathways of the
central nervous system because it does not cross the blood-brain barrier.
However, tryptophan and its metabolite 5-hydroxytryptophan (5-HTP), from which
serotonin is synthesized, can and do cross the blood-brain barrier. These
agents are available as dietary supplements and may be effective serotonergic
agents. One product of serotonin breakdown is 5-Hydroxyindoleacetic acid (5
HIAA), which is excreted in the urine. Serotonin and 5 HIAA are sometimes
produced in excess amounts by certain tumors or cancers, and levels of these
substances may be measured in the urine to test for these tumors.
The main functions of serotonin are: the regulation
of mood, appetite, sleep, muscle contraction, and some cognitive functions
including memory and learning. Modulation of serotonin at synapses is thought
to be a major action of several classes of pharmacological antidepressants.
Serotonin is a monoamine neurotransmitter synthesized in serotonergic neurons
in the central nervous system (CNS) and enterochromaffin cells in the
gastrointestinal tract. Serotonin is synthesized extensively in the human
gastrointestinal tract (about 90%), and the major storage place is platelets in
the blood stream.
In the central nervous system, serotonin is believed to play an
important role in the regulation of body temperature, mood, sleep, vomiting,
sexuality, and appetite. Low levels of serotonin have been associated with
several disorders, notably clinical depression, migraine, irritable bowel
syndrome, tinnitus, fibromyalgia, bipolar disorder, and anxiety disorders.
The
pharmacology of 5-HT is extremely complex, with its actions being mediated by a
large and diverse range of 5-HT receptors. At least seven different receptor
"families" are known to exist, each located in different parts of the
body and triggering different responses. As with all neurotransmitters, the
effects of 5-HT on the human mood and state of mind, and its role in
consciousness, are very difficult to ascertain. Serotonin (5-HT) receptors are
also used by other psychoactive drugs, including LSD, DMT, and psilocybin, the
active ingredient in psychedelic mushrooms.

The body
sometimes deliberately prolongs the transmission of a signal across a synapse
by slowing the destruction of neurotransmitters. It does this by releasing into
the synapse special long-lasting chemicals called neuromodulators. Some
neuromodulators aid the release of neurotransmitters into the synapse; others
inhibit the reabsorption of neurotransmitters so that they remain in the
synapse; still others delay the breakdown of neurotransmitters after their
reabsorption, leaving them in the tip to be released back into the synapse when
the next signal arrives.
Mood, pleasure, pain, and other mental states are determined by particular
groups of neurons in the brain that use special sets of neurotransmitters and
neuromodulators. Mood, for example, is strongly influenced by the
neurotransmitter serotonin. Many researchers think that depression results from
a shortage of serotonin. Prozac, the world’s bestselling antidepressant,
inhibits the reabsorption of serotonin, thus increasing the amount in the
synapse.

Drugs alter transmission of impulses across
the synapse.
Depression can result from a shortage of the neurotransmitter serotonin.
The antidepressant drug Prozac works by blocking reabsorption of serotonin in
the synapse, making up for the shortage.
Dopamine - an important transmitter
Arvid Carlsson
performed a series of pioneering studies during the late 1950's, which showed
that dopamine is an important transmitter in the brain. It was previously
believed that dopamine was only a precursor of another transmitter,
noradrenaline. Arvid Carlsson developed an assay that made it possible to
measure tissue levels of dopamine with high sensitivity. He found that dopamine
was concentrated in other areas of the brain than noradrenaline, which led him
to the conclusion that dopamine is a transmitter in itself. Dopamine existed in
particularly high concentrations in those parts of the brain, called the basal
ganglia, which are of particular importance for the control of motor behavior.
L-DOPA (L-3,4-dihydroxyphenylalanine) is a chemical that is made and used
as part of the normal biology of some animals and plants. Some animals
including humans make it via biosynthesis from the amino acid L-tyrosine.
L-DOPA is the precursor to the neurotransmitters dopamine, norepinephrine
(noradrenaline), and epinephrine (adrenaline) collectively known as
catecholamines. L-DOPA can be manufactured and in its pure form is sold as a
psychoactive drug with the
Dopamine is synthesized in the body from within
cells (mainly by neurons and cells in the medulla of the adrenal glands) and
can be created from any one of the following three amino acids:
L-Phenylalanine (PHE)
L-Tyrosine (L-4-hydroxyphenylalanine; TYR)
L-DOPA (L-3,4-dihydroxyphenylalanine; DOPA)
These amino acids are provided from natural sources such as the ingestion
of various kinds of food, with L-tyrosine being the most common of the three.
Although dopamine itself is also commonly found in many types of food, unlike
the amino acids that form it, it is incapable of crossing the protective blood-brain-barrier
(BBB), which severely restricts its functionality upon consumption. It must be
formed from within the walls of the BBB to properly perform its cognitive
duties, though not its peripheral actions. Dopamine itself is also used in the
synthesis of the following related catecholamine neurotransmitters:
Norepinephrine
(β,3,4-trihydroxyphenethylamine; Noradrenaline; NE, NA)
Epinephrine
(β,3-dihydroxy-N-methylphenethylamine; Adrenaline; EPI, ADR)
This is the
complete metabolic pathway:
L-Phenylalanine → L-Tyrosine →
L-DOPA → Dopamine → Norepinephrine → Epinephrine
L-Phenylalanine is converted into L-tyrosine by the enzyme phenylalanine
hydroxylase (PAH) with molecular oxygen (O2) and tetrahydrobiopterin (THB) as
cofactors. L-Tyrosine is converted into L-DOPA by the enzyme tyrosine
hydroxylase (TH) with tetrahydrobiopterin (THB), O2, and ferrous iron (Fe2+) as
cofactors. L-DOPA is converted into dopamine by the enzyme aromatic L-amino
acid decarboxylase (AAAD; also known as DOPA decarboxylase (DDC)) with
pyridoxal phosphate (PLP) as the cofactor. The reactions are illustrated as
follows:
PAH: L-Phenylalanine + THB + O2 + Fe2+ →
L-Tyrosine + DHB + H2O + Fe2+
TH: L-Tyrosine + THB + O2 + Fe2+ →
L-DOPA + DHFA + H2O + Fe2+
AAAD: L-DOPA + PLP → Dopamine +
PLP + CO2
Other decarboxylation reactions from the citric acid
cycle include:
to succinyl-CoA.
Drugs
against Parkinson's disease
Arvid Carlsson realized that the symptoms caused by reserpine were similar to
the syndrome of Parkinson's disease. This led, in turn, to the finding that
Parkinson patients have abnormally low concentrations of dopamine in the basal
ganglia. As a consequence L-dopa was developed as a drug against Parkinson's
disease and today still is the most important treatment for the disease. During
Parkinson's disease dopamine producing nerve cells in the basal ganglia
degenerate, which causes tremor, rigidity and akinesia. L-dopa, which is
converted to dopamine in the brain, compensates for the lack of dopamine and
normalizes motor behavior.
Antipsychotic
and antidepressive drugs
Apart from the successful treatment of Parkinson's disease Arvid Carlsson's
research has increased our understanding of the mechanism of several other
drugs. He showed that antipsychotic drugs, mostly used against schizophrenia,
affect synaptic transmission by blocking dopamine receptors. The discoveries of
Arvid Carlsson have had great importance for the treatment of depression, which
is one of our most common diseases. He has contributed strongly to the
development of selective serotonin uptake blockers, a new generation of
antidepressive drugs.
|
|
|
|
Dopamine nerve pathways in the brain.
Arvid Carlsson showed that there were particularly high levels of the chemical
transmitter dopamine in the so called basal ganglia of the brain, which are of
major importance for instance for the control of our muscle movements. In
Parkinson's disease those dopamine producing nerve cells whose nerve fibers
project to the basal ganglia die. This causes symptoms such as tremor, muscle
rigidity and a decreased ability to move about.
|
|
|
|
A message from one nerve cell to another is
transmitted with the help of different chemical transmitters. This occurs at
specific points of contact, synapses, between the nerve cells. The chemical transmitter
dopamine is formed from the precursors tyrosine and L-dopa and is stored in
vesicles in the nerve endings. When a nerve impulse causes the vesicles to
empty, dopamine receptors in the membrane of the receiving cell are influenced
such that the message is carried further into the cell. In the treatment of
Parkinson's disease, the drug L-dopa is given, and is converted to dopamine in
the brain. This compensates for the patient's lack of dopamine.
Sedatives
Sedatives (SED-uh-tivz), sometimes
called as tranquilizers (TRANK-will-LY-zerz) or sleeping pills, include
barbiturates or "downers." They are drugs that produce a calming
effect or sleepiness. Physicians prescribe them to relieve anxiety, promote
sleep, and treat seizures. When they are abused or taken at high doses,
however, many of these drugs can lead to loss of consciousness or even death.
Combining sedatives with alcohol is particularly dangerous. Possible effects of
sedative abuse include poor judgment, slurred speech, staggering, poor coordination,
and slow reflexes.

Cocaine
is a mood-altering drug that interferes with normal transport of the
neurotransmitter dopamine, which carries messages from neuron to neuron. When
cocaine molecules block dopamine receptors, too much dopamine remains active in
the synaptic gaps between neurons, creating feelings of excitement and
euphoria.
GABA - ![]() -aminobutyric acid
-aminobutyric acid
The term GABA refers to
the simple chemical substance ![]() -aminobutyric acid
(NH2CH2CH2 CH2COOH). It is the
major inhibitory neurotransmitter in the central nervous system.
Its presence in the brain first was reported in 1950 (Roberts and Frankel, 1950a).
-aminobutyric acid
(NH2CH2CH2 CH2COOH). It is the
major inhibitory neurotransmitter in the central nervous system.
Its presence in the brain first was reported in 1950 (Roberts and Frankel, 1950a).
For several years the presence of GABA in
brain remained a biochemical curiosity and a physiological enigma. It was
remarked in the first review written on GABA that “Perhaps the most difficult
question to answer would be whether the presence in the gray matter of the
central nervous system of uniquely high concentrations of ![]() -aminobutyric acid and the
enzyme which forms it from glutamic acid has a direct or indirect connection to
conduction of the nerve impulse in this tissue” . However, later that year, the
first suggestion that GABA might have an inhibitory function in the vertebrate
nervous system came from studies in which it was found that topically applied
solutions of GABA exerted inhibitory effects on electrical activity in the
brain. In 1957, the suggestion was made that indigenously occurring GABA might
have an inhibitory function in the central nervous system from studies with
convulsant hydrazides.
-aminobutyric acid and the
enzyme which forms it from glutamic acid has a direct or indirect connection to
conduction of the nerve impulse in this tissue” . However, later that year, the
first suggestion that GABA might have an inhibitory function in the vertebrate
nervous system came from studies in which it was found that topically applied
solutions of GABA exerted inhibitory effects on electrical activity in the
brain. In 1957, the suggestion was made that indigenously occurring GABA might
have an inhibitory function in the central nervous system from studies with
convulsant hydrazides.
Also in 1957, suggestive evidence for an
inhibitory function for GABA came from studies that established GABA as the
major factor in brain extracts responsible for the inhibitory action of these
extracts on the crayfish stretch receptor system. Within a brief period the
activity in this field increased greatly, so that the research being carried
out ranged all the way from the study of the effects of GABA on ionic movements
in single neurons
to clinical evaluation of the role of the GABA system in epilepsy,
schizophrenia, mental retardation, etc. This surge of
interest warranted the convocation in 1959 of the first truly interdisciplinary
neuroscience conference ever held, at which
were present most of the individuals who had played a role in opening up this
exciting field.

Schematic representation of the gamma-aminobutyric acid (GABAA)
receptor. The functional receptor consists of five proteins, or subunits--most
likely two α subunits, one β subunit, and two γ subunits. (Question marks indicate that the identity of these subunits
has not been confirmed.) The proposed binding sites for GABA (α and β subunits), benzodiazepines (adjacent α
and γ subunits), barbituates (unidentified subunit), and alcohol α, β, and γ subunits) are indicated. P's represent phosphate groups attached to the
receptor that regulate the receptor's activity and sensitivity to alcohol.
The trace amine
tryptamine is produced in the brain and possibly other tissues by the
decarboxylation of L-tryptophan. tryptamine is believed to function as a neurotransmitter
or neuromodulator in the CNS of mammals. It is catabolized by monoamine oxidase
A catalyzed oxidation to indole acetaldehyde. This aldehyde can then be
oxidized to indole-3-acetate by an undetermined isoform of aldehyde
dehydrogenase , or reduced to indole-3-ethanol (tryptophol) by aldehyde
reductase in the presence of NADPH. It was previously suggested that the
cytochrome P450 isozyme CYP2D6 mediates the oxidative deamination of
tryptamine, but later work showed that monoamine oxidase A was responsible for
this activity. An additional metabolite of tryptamine has been identified as
(4R)-2-(3-indolylmethyl)-1,3-thiazolidine-4-carboxylate, a condensation product
of indole acetaldehyde and free L-cysteine present in brain tissue. Its formation
in vitro is spontaneous and its bioligical function is unknown.
Histamine
Histamine
[2-(4-Imidazolyl)-ethylamine] is an important mediator of many biological
processes including inflammation, gastric acid secretion, neuromodulation, and
regulation of immune function. Due to its potent pharmacological activity even
at very low concentrations, the synthesis, transport, storage, release and
degradation of histamine have to be carefully regulated to avoid undesirable
reactions. Histamine is also generated by microbiological action in the course
of food processing and it is therefore present in substantial amounts in many
fermented foodstuffs and beverages.
Histamine
is formed by decarboxylation of the amino acid L-histidine in a reaction
catalyzed by the enzyme histidine decarboxylase. The major routes of histamine
inactivation in mammals are methylation of the imidazole ring, catalyzed by
histamine N-methyltransferase, and oxidative deamination of the primary amino
group, catalyzed by diamine oxidase.

Histidine
decarboxylase (HDC, EC 4.1.1.22) generates histamine by catalyzing the removal
of the carboxyl group from the amino acid L-histidine.
Mammalian
HDC enzymes utilize pyridoxal-phosphate as an active-site cofactor. HDC is
synthesized as a 74 kDa precursor consisting of 662 amino acid residues that is
processed to a 54 kDa active form that apparently forms homodimers. Although
the details of HDC processing have yet to be worked out the active enzyme
appears to be a cytosolic protein. In mammals, the enzyme is encoded by a
single gene with twelve exons designated HDC. The human HDC gene is located on
chromosome 15q21-22. HDC is an unstable protein that is synthesized only when a
cell needs to make histamine and the enzyme is immediately degraded when sufficient
histamine has been generated. Therefore, HDC is detectable only in cells
actively synthesizing histamine. In contrast, the histamine degrading enzymes
HMT and DAO are constitutively produced and are present in relatively constant
amounts. Mast cells, basophils, enterochromaffin-like cells in the gastric
mucosa and histaminergic neurons synthesize considerable amounts of histamine
and store the mediator in special storage granula inside the cell. Upon
appropriate stimulation, these cells can rapidly release relatively large
amounts of histamine and thereby efficiently activate suitable effector
mechanisms. In recent years, it became clear that apart from these
histamine-storing cell types, many other cells including epithelial cells and
lymphocytes can express HDC and synthesize histamine. In these cells, histamine
is not stored but appears to be made short-term in small amounts and
immediately released.
Key
Concepts:
Histamine
mediates a wide variety of physiological and pathological responses, such as
inflammation, gastric acid secretion, neurotransmission and immune modulation.
Histamine
is synthesised through decarboxylation of l‐histidine by histidine decarboxylase
and is catabolised through oxidative deamination or methylation.
Tissue
histamine levels are transiently increased by degranulation of mast cells and
basophils while they are upregulated by de novo synthesis in gastric
enterochromaffin‐like cells, and neurons.
Histamine
exerts its functions by acting on its specific receptors consisting of H1, H2,
H3 and H4 receptors.
Histamine
is a paracrine mediator, of which actions are generally limited in the local
microenvironment.
Histamine
H1 receptor antagonists have brought successful therapeutic approaches for immediate
allergy, because histamine evokes vasodilation and increased vascular
permeability by acting on the H1 receptor.
In
the central nervous system, histamine is involved in awakening, appetite,
maintenance of circadian rhythm, learning and memory, which are regulated by
the H1 and H3 receptors.
Histamine
stimulates parietal cells to induce gastric acid secretion by acting on the H2
receptor, of which antagonists drastically improved therapeutic approaches for
peptic ulcer.
Pre‐synaptic
histamine H3 receptor regulates release of various neurotransmitters including
histamine itself, and is expected as a potential drug target for the treatment
of cognitive dysfunctions.
Histamine
H4 receptor is expressed exclusively in blood cells and mediates their chemotaxis
in response to histamine.
Diagnostic
role of determination of AlAT and AsAT
Aspartate transaminase
Aspartate transaminase (AST),
also called aspartate aminotransferase (AspAT/ASAT/AAT) or serum
glutamic oxaloacetic transaminase (SGOT), is a pyridoxal phosphate (PLP)-dependent transaminase
enzyme. AST catalyzes the reversible transfer of an α-amino group between
aspartate and glutamate and, as such, is an important enzyme in amino acid
metabolism. AST is found in the liver, heart, skeletal muscle, kidneys,
brain,
and red blood cells, and it is commonly measured clinically as a marker for
liver health.
Function
Aspartate transaminase catalyzes the interconversion of aspartate
and α-ketoglutarate to oxaloacetate
and glutamate.
Aspartate (Asp) + α-ketoglutarate ↔
oxaloacetate + glutamate (Glu)
Reaction catalyzed by aspartate aminotransferase
As a prototypical transaminase, AST relies on PLP as a
cofactor to transfer the amino group from aspartate or glutamate to the
corresponding ketoacid.
In the process, the cofactor shuttles between PLP and the pyridoxamine phosphate (PMP) form. The
amino group transfer catalyzed by this enzyme is crucial in both amino acid
degradation and biosynthesis. In amino acid degradation, following the
conversion of α-ketoglutarate to glutamate, glutamate subsequently
undergoes oxidative deamination to form ammonium
ions, which are excreted as urea. In the reverse reaction, aspartate may be synthesized
from oxaloacetate, which is a key intermediate in the citric acid
cycle.
Isozymes
Two isoenzymes are present in a wide variety of
eukaryotes. In humans:
·
GOT1/cAST, the cytosolic
isoenzyme derives mainly from red blood
cells and heart.
·
GOT2/mAST, the mitochondrial
isoenzyme is present predominantly in liver.
These isoenzymes are thought to have evolved from a common ancestral AST
via gene duplication, and they share a sequence homology of approximately 45%.
AST has also been found in a number of microorganisms,
including E. coli,
H.
mediterranei, and T. thermophilus. In E. coli,
the enzyme is encoded by the aspCgene and has also been shown to exhibit
the activity of an aromatic-amino-acid transaminase.
Structure
Structure of aspartate transaminase from chicken heart
mitochondria
X-ray crystallography studies have been
performed to determine the structure of aspartate transaminase from various
sources, including chicken mitochondria, pig heart cytosol, and E. coli.
Overall, the three-dimensional polypeptide structure for all species is quite
similar. AST is dimeric, consisting of two identical subunits,
each with approximately 400 amino acid residues and a molecular weight of
approximately 45 kD. Each subunit is composed of a large and a small domain, as
well as a third domain consisting of the N-terminal residues 3-14; these few
residues form a strand, which links and stabilizes the two subunits of the
dimer. The large domain, which includes residues 48-325, binds the PLP cofactor
via an aldimine
linkage to the ε-amino group of Lys258. Other residues in this domain –
Asp 222 and Tyr 225 – also interact with PLP via hydrogen
bonding. The small domain consists of residues 15-47 and 326-410 and
represents a flexible region that shifts the enzyme from an "open" to
a "closed" conformation upon substrate binding.
The two independent active sites are positioned near the
interface between the two domains. Within each active site, a couple arginine
residues are responsible for the enzyme’s specificity for dicarboxylic
acid substrates: Arg386 interacts with the substrate’s proximal
(α-)carboxylate group, while Arg292 complexes with the distal (side-chain)
carboxylate.
In terms of secondary structure, AST contains both α
and β elements. Each domain has a central sheet of β-strands with
α-helices packed on either side.
Mechanism
Aspartate transaminase, as with all
transaminases, operates via dual substrate recognition; that is, it is able to
recognize and selectively bind two amino acids (Asp and Glu) with different
side-chains. In either case, the transaminase reaction consists of two similar
half-reactions that constitute what is referred to as a ping-pong mechanism. In the first
half-reaction, amino acid 1 (e.g., L-Asp) reacts with the enzyme-PLP complex to
generate ketoacid 1 (oxaloacetate) and the modified enzyme-PMP. In the second
half-reaction, ketoacid 2 (α-ketoglutarate) reacts with enzyme-PMP to
produce amino acid 2 (L-Glu), regenerating the original enzyme-PLP in the
process. Formation of a racemic product (D-Glu) is very rare.
The specific steps for the half-reaction
of Enzyme-PLP + aspartate ⇌ Enzyme-PMP + oxaloacetate are as
follows (see figure); the other half-reaction (not shown) proceeds in the
reverse manner, with α-ketoglutarate as the substrate.
Reaction
mechanism for aspartate aminotransferase
1.
Internal aldimine
formation: First, the ε-amino group of Lys258 forms a Schiff base
linkage with the aldehyde carbon to generate an internal aldimine.
2.
Transaldimination: The internal aldimine
then becomes an external aldimine when the ε-amino group of Lys258 is
displaced by the amino group of aspartate. This transaldimination reaction
occurs via a nucleophilic attack by the deprotonated amino
group of Asp and proceeds through a tetrahedral intermediate. As this point,
the carboxylate groups of Asp are stabilized by the guanidinium
groups of the enzyme’s Arg386 and Arg 292 residues.
3.
Quinonoid formation: The hydrogen attached
to the a-carbon of Asp is then abstracted (Lys258 is thought to be the proton
acceptor) to form a quinonoid intermediate.
4.
Ketimine
formation: The quinonoid is reprotonated, but now at the aldehyde carbon, to
form the ketimine intermediate.
5.
Ketimine hydrolysis:
Finally, the ketimine is hydrolyzed to form PMP and oxaloacetate.
This
mechanism is thought to have multiple partially rate-determining steps.[15]
However, it has been shown that the substrate binding step (transaldimination)
drives the catalytic reaction forward.
Clinical significance
AST is similar to alanine transaminase (ALT) in that both
enzymes are associated with liver parenchymal
cells. The difference is that ALT is found predominantly in the liver, with
clinically negligible quantities found in the kidneys, heart, and skeletal
muscle, while AST is found in the liver, heart (cardiac
muscle), skeletal muscle, kidneys, brain, and red blood cells. As a
result, ALT is a more specific indicator of liver inflammation
than AST, as AST may be elevated also in diseases affecting other organs, such
as myocardial infarction, acute pancreatitis, acute hemolytic
anemia, severe burns, acute renal
disease, musculoskeletal diseases, and trauma.
AST was defined as a biochemical marker
for the diagnosis of acute myocardial infarction in 1954. However, the use of
AST for such a diagnosis is now redundant and has been superseded by the cardiac
troponins.
AST (SGOT) is commonly measured
clinically as a part of diagnostic liver function tests, to determine liver
health.
|
Patient type |
|
|
Female |
6 - 34 IU/L |
|
Male |
8 - 40 IU/L |
Alanine transaminase
Alanine transaminase or ALT
is a transaminase
It is also called serum glutamic pyruvic transaminase (SGPT) or alanine
aminotransferase (ALAT).
ALT is found in serum
and in various bodily tissues, but is most commonly associated with the liver. It catalyzes the
two parts of the alanine cycle.
Function
It catalyzes the transfer of an amino group from alanine
to α-ketoglutarate, the products of this
reversible transamination reaction being pyruvate
and glutamate.
glutamate
+ pyruvate
⇌ α-ketoglutarate + alanine
Alanine
transaminase
ALT (and
all transaminases) require the coenzyme pyridoxal phosphate, which is converted into
pyridoxamine in the first phase of the reaction, when an amino acid is
converted into a keto acid.
Clinical significance
It is commonly measured clinically as a part of a diagnostic evaluation of hepatocellular injury, to determine liver
health. When used in diagnostics, it is almost always measured in international
units/liter (U/L). While sources vary on specific normal range values for
patients, 10-40 U/L is the standard normal range for experimental studies.
Alanine transaminase shows a marked diurnal
variation.
Elevated levels
|
Patient type |
|
|
Female |
5–38 IU/L |
|
Male |
10–50 IU/L |
Significantly elevated levels of
ALT(SGPT) often suggest the existence of other medical problems such as viral hepatitis,
diabetes,
congestive heart failure, liver damage, bile duct
problems, infectious mononucleosis, or myopathy.
For this reason, ALT is commonly used as a way of screening for liver problems.
Elevated ALT may also be caused by dietary choline deficiency. However,
elevated levels of ALT do not automatically mean that medical problems exist.
Fluctuation of ALT levels is normal over the course of the day, and ALT levels
can also increase in response to strenuous physical exercise.
When elevated ALT levels are found in the blood, the
possible underlying causes can be further narrowed down by measuring other
enzymes. For example, elevated ALT levels due to liver-cell
damage can be distinguished from biliary duct
problems by measuring alkaline phosphatase. Also, myopathy-related
ALT levels can be ruled out by measuring creatine
kinase enzymes. Many drugs may elevate ALT levels, including Zileuton,
omega-3-acid ethyl esters (Lovaza), anti-inflammatory drugs, antibiotics, cholesterol
medications, some antipscyhotics such as Risperidone, and anti-convulsants.
For years, the American Red Cross used ALT testing as part of
the battery of tests to ensure the safety of its blood supply by deferring
donors with elevated ALT levels. The intent was to identify donors potentially
infected with Hepatitis C because there was no specific test for that disease
at the time. Prior to July 1992, widespread blood donation testing in the
Enhanced
cardiac enzyme profile.
Abstract
A
protocol for an enhanced Cardiac Enzyme Profile is proposed based on an admission,
or initial, serum specimen and a second specimen 16 hours after onset of
symptoms as minimal baseline serum samples in order to accomplish several
simultaneous goals: 1. Detecting CK2MB at its average peak for maximal
assurance of diagnosis when release is small and for prognosis in all cases of
increased serum CK2MB 2. Detection of laboratory evidence of myocardial injury
when admission is delayed after onset by the collection of an admission sample
for declining CK2MB, and for assays of other enzymes with longer time curves
after myocardial injury such as LD isoenzymes and ASAT/ALAT activities and
ratio 3. Establishment of decision limits and criteria for the determination of
laboratory evidence of myocardial injury 4. Providing cost-effective procedures
other than limitation of the number of samples; these include establishing
thresholds and criteria for total CK, total LD, and ASAT so that isoenzymes and
ALAT are only performed when thresholds are exceeded and criteria are met;
performing only CK and, if the threshold is exceeded, CK isoenzymes on the
16-hour sample; collecting additional samples after the first two only when
indicated by positive or suspicious (borderline) results and only on routine
morning or afternoon rounds rather than specifically timed specimens (except in
cases involving thrombolytic therapy); and termination of the protocol once
peak positive CK2MB activity and requisite diagnostic consensus confirmation
(such as positive LD isoenzymes) is obtained whether or not thrombolytic
therapy is involved. Tissue localization of the enzymes has been outlined in
some detail with particular reference to the amount of CK2MB in skeletal
muscle. Pathophysiological factors discussed in more depth in a previous
article have been amplified here with particular reference to the role of
increased synthesis as a response to myocardial injury by surrounding
prehypertrophic and hypertrophic myocardium as a possible major source of
increased serum enzymes in myocardial infarction. ASAT and especially the
ASAT/ALAT ratio are useful tests in the protocol, particularly in cases tested
late after onset of symptoms when CK2MB has declined into the borderline or
usual range, and ASAT/ALAT may be helpful in evaluating LD isoenzyme results.
Codes for interpretive comments are provided to serve as guidelines.
Use of enzymes for the diagnosis of alcohol-related organ
damage.
Abstract
Elevated
levels of serum enzymes are frequently associated not only with alcohol-related
organ damage but also with excessive alcohol consumption and alcoholism without
significant tissue injury. However, both in the early detection of alcoholism
as well as also in the diagnosis of alcohol-related diseases the sensitivities
and specificities of these enzyme markers vary considerably. They may be
influenced by nonalcohol-related diseases, enzyme-inducing drugs, nutritional
factors, metabolic disorders, age, smoking, etc. Consequently, we have neither
a single laboratory test--enzyme marker--nor a test combination that is
reliable enough for the exact diagnosis between alcohol- and nonalcohol-related
organ damage. In most cases it is possible to determine the tissue from which
the elevated enzyme is derived, but only occasionally enzyme changes reflect
the quantity of the tissue injury. Gamma-glutamyltransferase (GGT) is the most
widely used laboratory marker of alcoholism and heavy drinking, detecting
34-85% of problem drinkers and alcoholics. However, the unspecificity of
increased serum GGT limits its use for general screening purposes. Its value in
the follow-up of various treatment programs, however, is well established. An
elevated level of serum aspartate aminotransferase (ASAT) and alanine
aminotransferase (ALAT) in an alcoholic or a heavy consumer indicates
alcohol-induced organ damage. The use of test combinations significantly
improves the information received with single serum enzyme determinations. An
ASAT/ALAT ratio greater than 1.5 can be considered as highly suggestive for the
alcoholic etiology of the liver injury. Still better discrimination between
alcoholic and nonalcoholic origin of the liver disease may be achieved by the
determination of the ratio of GGT to alkaline phosphatase. If this ratio
exceeds 1.4 the specificity of the finding in favor for alcoholic liver injury
is 78%. The determination of the mitochondrial isoenzyme of ASAT also improves
the diagnostic value of ASAT determination. The ratio of mitochondrial
isoenzyme to total over 4 is highly suggestive for alcohol-related liver
injury. In general, however, the determination of serum activities of other
enzymes such as ornithine carbamyl transferase, lactate dehydrogenase,
isocitrate dehydrogenase, sorbitol dehydrogenase, alcohol dehydrogenase,
guanase, aldolase, alkaline phosphatase or glutathione S-transferase do not
significantly improve the diagnostic information obtained with more
conventional laboratory markers of liver injury.






























