Non-protein nitrogenous containing and nitrogen
not containing organic components of blood. Residual nitrogen. Lipoproteins of blood plasma.
Biochemistry of immune
processes and boichemical mechanisms of
immunodeficit states.
Residual nitrogen, its components, ways of their
formation, blood content
The
state of protein nutrition can be determined by measuring the dietary intake
and output of nitrogenous compounds from the body. Although nucleic acids also
contain nitrogen, protein is the major dietary source of nitrogen and
measurement of total nitrogen intake gives a good estimate of protein intake
(mg N Ч 6.25 = mg protein, as nitrogen is 16% of most proteins). The output of nitrogen
from the body is mainly in urea and smaller quantities of other compounds in
urine and undigested protein in feces, and significant amounts may also be lost
in sweat and shed skin.
The
difference between intake and output of nitrogenous compounds is known as nitrogen
balance. Three states can be defined: In a healthy adult, nitrogen balance
is in equilibrium when intake equals output, and there is no change in
the total body content of protein. In a growing child, a pregnant woman, or in
recovery from protein loss, the excretion of nitrogenous compounds is less than
the dietary intake and there is net retention of nitrogen in the body as
protein, ie, positive nitrogen balance. In response to trauma or
infection or if the intake of protein is inadequate to meet requirements there
is net loss of protein nitrogen from the body, ie, negative nitrogen
balance. The continual catabolism of tissue proteins creates the
requirement for dietary protein even in an adult who is not growing, though
some of the amino acids released can be reutilized.
Nitrogen
balance studies show that the average daily requirement is
Residual nitrogen –
nonprotein nitrogen, that is nitrogen of organic and inorganic compounds that
remain in blood after protein sedimentation.
Organic and inorganic compounds of residual
nitrogen are as follows: urea (50 % of the residual nitrogen), amino acids (25
%), creatine and creatinine (7,5 %), salts of ammonia and indicane (0,5 %),
other compounds (about 13 %).
Urea is formed in liver during the degradation of amino acids,
pyrimidine nucleotides and other nitrogen containing compounds. Amino acids are
formed as result of protein decomposition or owing to the conversion of fatty
acids or carbohydrates to amino acids. The pool of amino acids in blood is also
supported by the process of their absorption in intestine. Creatine is produced
in kidneys and liver from amino acids glycine and arginine, creatinine is
formed in muscles as result of creatine phosphate splitting. In result of
ammonia neutralization the ammonia salts can be formed. Indicane is the product
of indol neutralization in the liver.

Creatinine
Urine
The
content of residual nitrogen in blood is 0,2 – 0,4 g/l.
The pathways of convertion of amino acid
nonnitrogen residues.
The removal of the amino
group of an amino acid by transamination or oxidative deamination produces an
α-keto acid that contains the carbon skeleton from the amino acid (nonnitrogen residues). These α-keto
acids can be used for the biosynthesis of non-essential amino acids or
undergoes a different degradation process. For alanine and serine, the
degradation requires a single step. For most carbon arrangements, however,
multistep reaction sequences are required.
There are only seven degradation sequences for 20 amino acids. The seven
degradation products are pyruvate, acetyl CoA, acetoacetyl CoA, α-ketoglutarate,
succinyl CoA, fumarate, and oxaloacetate. The last four products are
intermediates in the citric acid cycle. Some amino acids have more than one
pathway for degradation.
The major point of entry into the tricarboxylate cycle is via
acetyl-CoA; 10 amino acids enter by this route. Of these, six (alanine, glycine,
serine, threonine, tryptophan and cysteine) are degraded to acetyl-CoA via
pyruvate, five (phenylalanine, tyrosine,
leucine, lysine, and tryptophan) are degraded via acetoacetyl-CoA, and
three (isoleucine, leucine and tryptophan)
yield acetyl-CoA directly. Leucine and
tryptophan yield both acetoacetyl-CoA and acetyl-CoA as end products.
The carbon skeletons of five amino acids (arginine, histidine, glutamate, glutamine and proline) enter the
tricarboxylic acid cycle via a-ketoglutarate.
The carbon skeletons of methionine,
isoleucine, and valine are ultimately degraded via propionyl-CoA and
methyl-malonyl-CoA to succinyl-CoA; these amino acids are thus glycogenic.
Fumarate is formed in catabolism of phenylalanine,
aspartate and tyrosine.
Oxaloacetate is formed in catabolism of aspartate and asparagine. Aspartate is
converted to the oxaloacetate by transamination.
Amino
acids that are degraded to citric acid cycle intermediates can serve as glucose
precursors and are called glucogenic. A glucogenic amino acid is an
amino acid whose carbon-containing degradation product(s) can be used to
produce glucose via gluconeogenesis.
Amino acids that are
degraded to acetyl CoA or acetoacetyl CoA can contribute to the formation of
fatty acids or ketone bodies and are called ketogenic. A ketogenic amino acid is an amino acid whose carbon-containing
degradation product(s) can be used to produce ketone bodies.
Amino acids that are degraded to pyruvate can be
either glucogenic or ketogenic. Pyruvate can be metabolized to either
oxaloacetate (glucogenic) or acetyl CoA (ketogenic).
Only two amino acids are purely ketogenic:
leucine and lysine. Nine amino acids are both glucogenic and ketogenic:
those degraded to pyruvate (alanine, glycine, cysteine, serine, threonine,
tryptophan), as well as tyrosine, phenylalanine, and isoleucine (which have two
degradation products). The remaining nine amino acids are purely glucogenic
(arginine, asparagine, aspartate, glutamine, glutamate, valine, histidine, methionine, proline)
The
regulation of protein metabolism. Protein metabolism is
regulated by different hormones. All hormones according to their action on
protein synthesis or splitting are divided on two groups: anabolic and
catabolic. Anabolic hormones promote to the protein synthesis. Catabolic
hormones enhance the decomposition of proteins.
Somatotropic hormone (STH,
growth hormone):
-
stimulates the passing of amino acids
into the cells;
- activates the synthesis of proteins, DNA,
RNA.
Thyroxine and triiodthyronine:
-
in
normal concentration stimulate the synthesis of proteins and nucleic acids;
-
in
excessive concentration activate the catabolic processes.
Insulin:
-
increases the permeability of cell
membranes for amino acids;
-
activates synthesis of proteins and
nucleic acids;
-
inhibits the conversion of amino
acids into carbohydrates.
Glucagon:
-
stimulates
the conversion of amino acids into carbohydrates.
Epinephrine:
- activates the
protein decomposition.
Glucocorticoids:
-
stimulate the catabolic
processes (protein decomposition) in connective, lymphoid and muscle tissues
and activate the processes of protein synthesis in liver;
-
stimulate the activity of
aminotransferases;
-
activate the synthesis of urea.
Sex hormones:
-
stimulate the processes of
protein, DNA, RNA synthesis;
-
cause the positive nitrogenous
balance.
The
role of liver in protein metabolism:
–
synthesis of plasma proteins. Most
of plasma proteins are synthesized in liver: all albumins, 75-90 % of
α-globulins, 50 % of β-globulins, all proteins of blood clotting
systems (prothrombin, fibrinogen, proconvertin, proaccelerine). Only
γ-globulins are synthesized in the cells of reticuloendothelial system.
–
synthesis of urea and uric acid;
–
synthesis of choline and creatine;
–
transamination and deamination of
amino acids.
Clinical
significance of residual nitrogen measurement in blood. The kinds of azotemia.
Azotemia
- increase of
the residual nitrogen content in blood. There are two kinds of azotemia: absolute and relative.
Absolute azotemia – accumulation of the
components of residual nitrogen in blood. Relative
azotemia occurs in dehydration of the organism (diarrhea, vomiting).
Absolute azotemia can be divided on the productive azotemia and retention azotemia. Retention azotemia is
caused by the poor excretion of the nitrogen containing compounds via the
kidneys; in this case the entry of nitrogen containing compounds into the blood
is normal.
Retention
azotemia can be divided on the renal and extrarenal. Renal retention azotemia occurs in kidney
diseases (glomerulonephritis, pyelonephritis, kidney tuberculosis et c.). Extrarenal retention azotemia is caused
by the violations of kidney hemodynamic and decrease of glomerulus filtration
processes (heart failure, local disorders of kidney hemodynamic).
Productive azotemia
is conditioned by the enhanced entry of
nitrogen containing compounds into the blood. The function of kidneys in this
case doesn’t suffer. Productive azotemia can be observed in cachexia,
leukoses, malignant tumors, treatment by glucocorticoids.
Prerenal Azotemia
Alternate
Names : Azotemia -
Prerenal, Renal Underperfusion, Uremia
Kidney Anatomy
Azotemia
From Wikipedia, the free encyclopedia
Jump to:
navigation,
search
It has
been suggested that this article or section be merged into uremia. (Discuss)
Azotemia is a
medical condition characterized by abnormal levels of urea, creatinine,
various body waste compounds, and other nitrogen-rich compounds in the blood as a result of
insufficient filtering of the blood by the kidneys.
Uremia can be used
as a synonym, or can be used to indicate severe azotemia, in which symptoms are
produced.
Azotemia
can be classified according to its cause. In prerenal azotemia the blood
supply to the kidneys is inadequate. In postrenal azotemia the urinary
outflow tract is obstructed. Other forms of azotemia are caused by diseases of
the kidneys themselves.
Other
causes of azotemia include congestive heart failure, shock, severe
burns, prolonged vomiting or diarrhea, some antiviral medications, liver
failure, or trauma to the kidney(s).
[edit] Signs and symptoms (prerenal azotemia)
- Decreased or absent urine output
- Fatigue
- Decreased alertness
- Confusion
- Pale skin color
- Rapid pulse
- Dry mouth
- Thirst, swelling (edema, anasarca)
- Orthostatic blood pressure (rises
or falls, significantly depending on position)
A urinalysis
will typically show a decreased urine sodium level, a high urine creatinine-to-
serum creatinine ratio, a high urine urea-to-serum urea ratio, and concentrated
urine (determined by osmolality and specific gravity). None of these is
particularly useful in diagnosis.
Prompt
treatment of some causes of azotemia can result in restoration of kidney
function; delayed treatment may result in permanent loss of renal function.
Treatment may include hemodialysis or peritoneal dialysis,
medications to increase cardiac output and increase blood pressure, and the
treatment of the condition that caused the azotemia to begin with. NOTE:
Azotemia is not diagnosed with abnormally high levels of Creatinine. Azotemia
simply refers to an elevated level of urea in the blood.
Added
Note: Uremia is not azotemia. Azotemia is one of many
clinical characteristics of uremia, which is a syndome characteristic of renal
disease. Uremia includes Azotemia, as well as acidosis, hyperkalemia,
hypertension, anemia and hypocalcemia along with other findings.
Retrieved
from "http://en.wikipedia.org/wiki/Azotemia"
PATIENT
HISTORY:
The patient
is a 60 year old male with a previous history of thoracic aortic aneurysm.
Currently presents with an abdominal aortic aneurysm and liver and kidney
masses. Admitted for
liver and kidney transplant.
The
specimen is received unfixed and in three parts.
Part I:
Part 1 is
labeled "liver" and consists of a
Part II:
Part 2 is
labeled "left kidney" and consists of a
Part III:
Part 3 is
labeled "spleen" and consists of a
MICROSCOPIC
DESCRIPTION:
Sections from
the liver show extensive cyst formation affecting more than 90% of the liver
parenchyma. The limited amount of liver tissue which remains shows a variety of
changes varying from atrophy to hemorrhage. Similar epithelium lined cysts are
seen in the kidney. Some cysts have ruptured and lead to hemorrhagic necrosis,
calcification and fibrosis. Focal cholesterol clefts and foreign body giant
cells are seen. The spleen shows an expanded red pulp and multiple cystic
spaces lined by flattened cells.
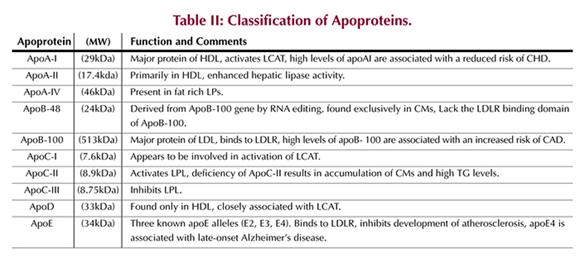
Lipoproteins and Apoproteins
http://www.youtube.com/watch?v=97uiV4RiSAY
Lipids are a
group of fatty substances that includes triglycerides (fat), phospholipids and
sterols (e.g. cholesterol). They constitute an important source of
energy, serve as precursors for a number of essential compounds, and are key
components of cells and tissues. Cholesterol, for example, is an
indispensable constituent of cellular membranes (1), as
well as the precursor for both steroid hormones and bile acids. On
average, the body utilizes approximately 1000 milligrams of cholesterol per
day, 30% of which comes directly from foods of animal origin, and the rest is
synthesized in the liver. Due to the insolubility of cholesterol and other
fatty compounds in the blood, their redistribution in the body requires
specialized carriers capable of solubilzing, ferrying, and unloading them at
specific target sites. Miscarriage of lipids while in circulation may lead to
atherosclerosis; a clinical condition marked by fatty deposits in the inner
walls of arteries, and the leading cause of death and disability in Western
countries.
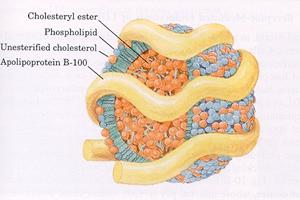
Most lipids are
transported in the blood as part of soluble complexes called lipoproteins
(LPs). Plasma LPs are spherical particles composed of a hydrophobic lipid core surrounded
by a hydrophilic layer, which renders the particles soluble. The lipid core
contains primarily triglycerides (TG) and cholesteryl esters (CE), as well as
small amounts of other fatty compounds, such as sphingolipids and fat-soluble
vitamins (e.g. vitamins A, D, E, and K). The external layer is made of
phospholipids, unesterified cholesterol, and specialized proteins, called
apolipoproteins or apoproteins. These proteins facilitate lipid solubilization
and help to maintain the structural integrity of LPs. They also serve as
ligands for LP receptors and regulate the activity of LP metabolic enzymes. As
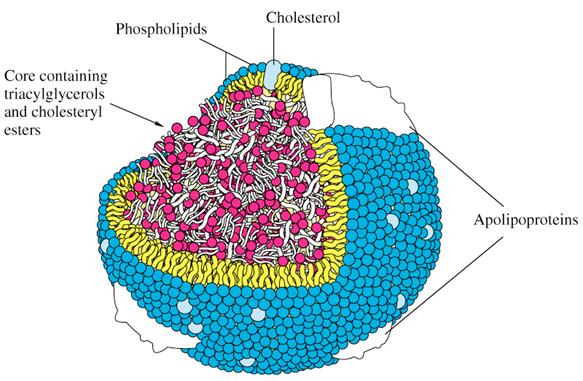
depicted in (Figure 1), the amphipathic molecules that compose the
outer layer of LPs are arranged so that their hydrophobic parts face the
central core, and their hydrophilic regions face the surrounding aqueous
environment.
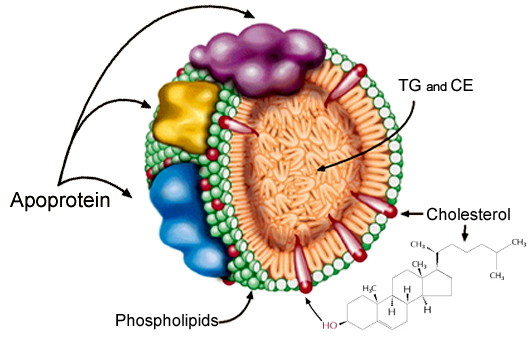
Figure 1: Schematic
Illustration of a Lipoprotein Particle
Cholesteryl
esters, which do not contain a free hydroxyl group (-OH) are more hydrophobic
than cholesterol, and better accommodated in the core of LPs. The conversion of
cholesterol to CE is catalyzed by a LP-associated enzyme called
lecithin-cholesterol acyltransferase (LCAT). This enzyme, which promotes
packaging of cholesteryl molecules in LPs, is critical for normal cholesterol
metabolism. Deficiency of LCAT activity leads to accumulation of unesterified
cholesterol in tissues, and is associated with a number of clinical conditions
including corneal opacity, hemolytic anemia, and premature atherosclerosis.
During ordinary metabolism,
plasma LPs lose, acquire, and exchange their lipid and protein constituents.
Normally, fat-rich LPs lose most of their fat within a few hours of food
ingestion, and become smaller and denser particles with higher relative
cholesterol content. The depletion of fat from LPs is catalyzed by lipoprotein
lipase (LPL). This lipolytic enzyme is located on the surface of endothelial
capillaries, and degrades triglycerides to free fatty acids (FFAs) and
glycerol. The released FFAs may stay in circulation bound to albumin, or be
taken-up by muscle and fat cells for usage and storage, respectively.
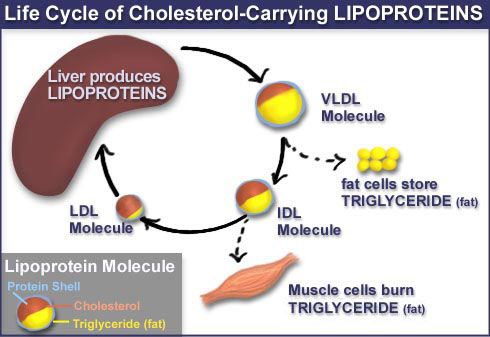
Lipids of dietary origin are
processed by intestinal epithelial cells, and then secreted into the
bloodstream as part of large, fat-rich LPs called chylomicrons (chylo = milky,
micron= indicates particle size). En route to the liver, chylomicrons
(CM) pass through endothelial capillaries, lose some fat, and their remnants
are taken-up by liver cells. In the liver, the lipids obtained from CM remnants
are re-processed and then secreted back into the bloodstream as part of very
low-density LPs (VLDL). Depletion of fat from VLDL transforms the particle into
an intermediate density lipoprotein (IDL), which upon further degradation of
its fat is converted into a relatively stable particle, called low density
lipoprotein (LDL). Because of its high cholesterol content, LDL is also called
LDL-cholesterol. Of the total blood cholesterol, 60-75% is found in LDL and the
rest primarily in high-density lipoprotein (HDL) particles. The main
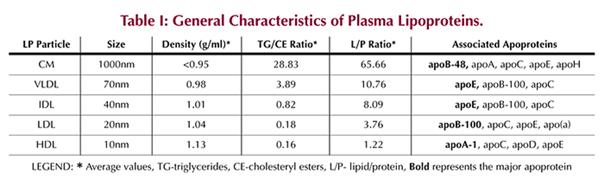
characteristics of plasma LPs and their associated apoproteins are summarized
in (Tables I and II),
respectively.
All peripheral
cells express the LDL-receptor (LDLR), and recycle it to the cell surface upon
need for cholesterol. Cholesterol is delivered to these cells through binding
of LDL to LDLR, which triggers endocytosis (internalization) of both species.
When the need for cholesterol is satisfied, the recycling of LDLR is
discontinued. Normally, an LDL particle stays in circulation for no more
than a few days before being consumed by a cholesterol needing cell. However,
under conditions of sustained cholesterol excess, the particle stays in
circulation for longer periods of time, and becomes more vulnerable to
undesired modifications (e.g. oxidation). As high levels of oxidized LDL are
commonly found in atherosclerotic plaques, they are thought to be the major
inducer of atherosclerotic lesions. Hence, LDL became known as bad cholesterol.
However, today we know that not all LDL particles are bad, and that some LDL
particles, especially very large ones (with diameter >21.3nm), may even
provide protection against atherosclerosis (2). LDL and HDL particle sizes are largely determined by a LP-associated
protein, called CETP (cholesteryl ester transfer protein). This protein
enhances exchange of non-polar lipids, primarily CE and TG, and facilitates
tight packaging of CE within the core of the particles. The end result of
prolonged and/or efficient CETP action is smaller LDL and HDL particles. [The
LP-anchored CETP can be envisioned as having a hand that rotates between the
interior and exterior of the particle and capable of holding only one
lipid molecule at a time. Grasping of one molecule releases another and vise
versa.]
Genetic variation at the human
CETP gene generates proteins with varying degrees of activity. For
example, a single codon variation, from isoleucine to valine at position 405,
generates a mutant protein, designated I405V, which manifests significantly
reduced CETP activity (3,
4). In a new observational study,
Barzilai, N. et al. (2) found that people with homozygosity for
the I405V allele have larger HDL and LDL particles, and that this genotype is associated
with exceptional longevity and a markedly reduced risk of coronary artery
disease (CAD). Of the 213 centenarians enrolled in the study, 80% had a high
proportion of large LDL particles, compared to just 8% of the subjects in the
control group (256 people in their 60’s and 70’s) (2).
Interestingly, HDL and LDL particle sizes are significantly larger in women
than in men, which may account, at least in part, for the longer life
expectancies of women.
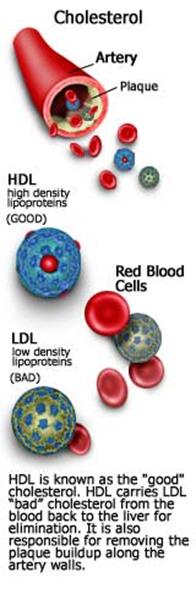
Unlike LDL, HDL is not recognized
by LDLR, and cannot deliver cholesterol to tissue cells. Instead, it has the
ability to remove excess peripheral cholesterol and return it to the liver for
recycling and excretion. This process, called reverse cholesterol transport, is
thought to protect against atherosclerosis. Observational studies over the last
2 decades have consistently shown strong correlation between elevated HDL
levels and low incidents of coronary heart disease (CHD). Hence HDL has been
dubbed “good” cholesterol.
HDL is synthesized in the liver and
intestine as a nascent, discoid-shaped particle that contains predominantly
apoA-I, and some phospholipids. Upon maturation, HDL assumes a spherical shape,
and the composition of its core lipids becomes very similar to that of LDL.
However, the relative higher protein content in HDL renders the particle denser
and more resistant to undesired modifications. Unlike the case of LDL, the
clearance of HDL from circulation is not negatively affected by excess
cholesterol, which may be another reason why HDL, despite being much smaller
particle than LDL (10nm versus 20nm), is not found in atherosclerotic plaques.
It’s worth noting, that the potential of LPs to become harmful is also
influenced by the character of their lipid constituents. For example, vitamin E
and lipids containing omega-3 fatty acid moieties appear to protect the
particles from harmful oxidation and from getting stuck on the walls of blood
vessels.
The functional difference between
LDL and HDL results primarily from the different character of their major
apoproteins, apoB-100 and apoA-I, respectively. ApoB-100, which is found
in VLDL, IDL, and LDL, but not in HDL, serves as a ligand for LDLR, and
provides LDL with the means to deliver cholesterol to tissue cells. On the
other hand, apoA-I, which is found exclusively in HDL, has a unique ability to
capture and solubilze free cholesterol. This apoA-I ability enables HDL to act
as a cholesterol scavenger.
A mutant apoA-I protein, called
apoA-I Milano (apoA-Im), has been identified in a group of people that live in
a small village in northern Italy (5).
Carriers of this protein, all heterozygous for the mutation, had very low
levels of HDL (7-14 mg/dl) but showed no clinical signs of
atherosclerosis (5-7). HDL particles in these subjects were markedly
larger than control (12nm versus 9.4nm), which may account for their immunity
against premature atherosclerosis. ApoA-Im differs from natural apoA-I by
having a cysteine residue at position 173 instead of arginine. This cysteine
residue forms disulfide bridges with other apoA-I molecules or with
apoA-II (6, 7), which apparently lead to larger HDL particles.
It also renders apoA-I more susceptible to catabolism (8), accounting for the low HDL levels in apoA-Im carriers.
The therapeutic potential of
apoA-I has been recently assessed in patients with acute coronary
syndromes (9). Of the 47 patients that participated in a
randomized controlled trial, 36 received 5 weekly infusions of recombinant
apoA-Im/phospholipid complexes, and 11 received only saline infusions. The
results showed significant regression in coronary atherosclerotic volume in the
apoA-Im treated group, and virtually no change in the control group (9). These results, if reproduced in larger clinical trials, may constitute
a revolutionary breakthrough in the non-invasive treatment of cardiovascular
disease. They should also encourage further exploration into the therapeutic
usefulness of apoA-Im and normal apoA-I in managing atherosclerotic vascular
diseases.
e LDL (low density lipoproteins) and HDL
(high density lipoproteins).
|
|
Lipid Levels
The lilipoproteins - Any of the series of soluble lipid-protein
complexes which are transported in the blood; each aggregate particle consists of
a spherical hydrophobic core containing triglycerides and cholesterol esters
surrounded by an amphipathic monolayer of phopholipids, cholesterol and
apolipoproteins; classes of lipoproteins include chylomicrons, very low-density
lipoproteins (VLDL), intermediate-density lipoproteins (IDL), low-density
lipoproteins (LDL), and high-density lipoproteins (HDL).
chylomicrons -
The class of largest diameter soluble lipid-protein complexes which the lowest in
density (mass to volume ratio); their composition is ~2% apolipoproteins, ~5%
cholesterol, and ~93% triglycerides and phospholipids; their normal role is to
be synthesized by the intestinal mucosal cells to transport dietary (exogenous)
triglycerides and other lipids from the intestines via the lacteals and
lymphatic system to the systemic circulation to the adipose tissue and liver
for storage and use; they are only present in the blood in significant
quantities after the digestion of a meal.
low-density lipoproteins (LDL) -
The class of large diameter soluble lipid-protein complexes which the fourth
lowest in density (mass to volume ratio); their composition is ~25%
apolipoproteins, ~45% cholesterol, and ~30% triglycerides and phospholipids;
their normal role is to transport cholesterol and other lipids from the liver
and intestines to the tissues for use; elevated levels of LDL are associated
with increased risk of cardiovascular disease. nickname - bad cholesterol
high-density lipoproteins (HDL) -
The class of small diameter soluble lipid-protein complexes which the highest
in density (mass to volume ratio); their composition is ~45% apolipoproteins,
~25% cholesterol, and ~30% triglycerides and phospholipids; their normal role
is to transport cholesterol and other lipids from the tissues to the liver for
disposal; elevated levels of HDL are associated with decreased risk of
cardiovascular disease.
very low-density lipoproteins (VLDL) -
The class of very large diameter soluble lipid-protein complexes which the
second lowest in density (mass to volume ratio); their composition is ~10%
apolipoproteins, ~40% cholesterol, and ~50% triglycerides and phospholipids;
their normal role is to transport triglycerides and other lipids from the liver
and intestines to the tissues for use; elevated levels of VLDL are associated
with some increased risk of cardiovascular disease.
http://www.youtube.com/watch?v=97uiV4RiSAY
What is
Cholesterol?
Cholesterol is a waxy fat
found in the body and, despite what you may have been told, is a necessary
nutrient for the body. Cholesterol is used in the formation of cell membranes
and plays an important role in hormone, bile and vitamin D production.
Cholesterol comes from two sources: the foods that we eat, such as meat, dairy
products and eggs, and our own liver, which produces about eighty percent of
all the cholesterol in the body. That means that only about twenty percent of
our total cholesterol is obtained from food. Since cholesterol is not
water-soluble, the liver packages the cholesterol into tiny spheres called
lipoproteins so that the cholesterol can be transported through the blood. The
lipoproteins can be divided into two different categories: low density and high
density lipoproteins.
http://www.youtube.com/watch?v=-WhADd1GKtA&feature=relmfu
Low density
lipoprotein (LDL): LDL, often dubbed the
"bad" cholesterol, carries most of the cholesterol in the blood and
seems to play a role in the deposition of fat in arteries. These deposits
result in blockages called plaque. In addition to narrowing the
arteries and increasing blood pressure, plaque contributes to the hardening of
artery walls, a condition known as atherosclerosis.

High density
lipoprotein (HDL): HDL is known as the
"good" cholesterol. HDL carries cholesterol from the blood back to
the liver for elimination. It is also responsible for removing the plaque
buildup along the artery walls. Elevated levels of HDL are very desirable
because it helps to clear blockages in the arteries, reduces LDL and decreases
blood pressure.
What are
Triglycerides?
Triglycerides are lipids
normally found in increased levels in the blood following the digestion of fats
in the intestine. Consumed calories that are not immediately used are stored in
fat cells in the form of triglycerides and are later released from fatty
tissues when the body needs energy between meals. The major transporter of
triglycerides is a forerunner of LDL, a simpler molecule known as VLDL
(very low density lipoprotein). As the VLDL loses triglycerides, the VLDL
particle is converted into intermediate and then low density lipoprotein. Over
time, elevated triglyceride levels may result in pancreatitis—a condition that can
cause malabsorption of nutrients and lead to diabetes. As pancreatitis
progresses, damage can spread to other organs, including the heart, lungs and
kidneys. High triglyceride levels also promote the deposition of cholesterol in
the arteries and are associated with known risk factors for heart disease. The
exact role that triglycerides play as an independent risk factor is not yet
clear because people with high LDL and low HDL levels also have high
triglyceride levels.
Although These
Researchers Beg to Differ…
One study by Koren-Morag,
Graff and Goldbourt, published in the American Heart Association journal Circulation,
found that individuals with elevated triglyceride levels have a nearly thirty
percent increased probability of suffering a stroke, even after taking into
account other risk factors such as cholesterol levels. One of the most
important aspects of the study is that it clarifies the independent link of
triglyceride levels to stroke, meaning that a causal relationship is likely.
What is Plaque?
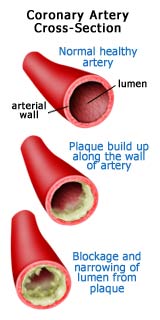
Excess LDL cholesterol clings to arterial walls as it is transported through the system. Macrophages eat the LDL and become "foam cells." The cells eventually rupture and begin to form a lipid layer called plaque. Connective fibers form in and around the fatty layer, causing it to harden. Over time, the fibrous layer thickens, narrowing the arterial pathway. When calcium deposits form a crust, the plaque becomes brittle and is more likely to rupture.
The Problem With Plaque
High blood cholesterol levels increase the
likelihood that the fat will be deposited as plaque on the inner surface of
arterial walls. As these deposits increase, the channel of the artery narrows,
contributing to an increase in blood pressure. To compensate, the heart must
work harder to pump the same volume of blood through the narrower arteries.
When the coronary arteries themselves are affected by plaque, the harder
working heart receives less oxygen, thus increasing the risk of heart attack.
Plaque also contributes to hardening of the arteries, or atherosclerosis.
This loss of flexibility in arterial walls elevates blood pressure, putting the
heart at additional risk. When the plaque deposits become unstable, they burst,
releasing their cholesterol into the bloodstream all at once. This can trigger
clotting in small coronary arteries. When the artery is completely obstructed,
blood flow stops and a heart attack occurs.
http://www.youtube.com/watch?v=XLLBlBiboJI&feature=related
http://www.youtube.com/watch?v=-WhADd1GKtA&feature=relmfu
What is a Lipoprotein?
Lipids, such as triacylglycerols and cholesterol
esters, are virtually insoluble in aqueous solution. Therefore, lipids must be
transported by the circulation in COMPLEX WITH water-soluble PROTEINS.
This complex LIPOPROTEIN is a globular micelle-like
particle that consists of a nonpolar core of triacylglycerols and cholesterol
esters surrounded by an amphiphilic coating of protein, phospholipid, and
cholesterol.
Here is a diagram of Low-Density Lipoprotein (LDL)
which is approximately 25nm in diameter:
http://www.youtube.com/watch?v=x-4ZQaiZry8
You need "Quick Time" Player and
Plug-In to view this LDL particle in motion:
http://www.youtube.com/watch?v=97uiV4RiSAY
Characteristics of Lipoproteins in Human Plasma
|
Characteristic |
Chylomicrons |
VLDL |
IDL |
LDL |
HDL |
|
Density (g/cm) |
~0.95 |
~1.006 |
1.006-1.019 |
1.019-1.063 |
1.063-1.210 |
|
Particle Diameter (nm) |
75-1200 |
30-80 |
25-35 |
18-25 |
5-12 |
|
Particle Mass (kD) |
400,000 |
10,000-80,000 |
5000-10,000 |
2300 |
175-360 |
|
%Proteina |
1.5-2.5 |
5-10 |
15-20 |
20-25 |
40-55 |
|
%Phospholipidsa |
7-9 |
15-20 |
22 |
15-20 |
20-35 |
|
%Free Cholesterola |
1-3 |
5-10 |
8 |
7-10 |
3-4 |
|
%Triacylglycerolsb |
84-89 |
50-65 |
22 |
7-10 |
3-5 |
|
%Cholesteryl Estersb |
3-5 |
10-15 |
30 |
35-40 |
12 |
|
Major Apolipoproteins |
AI,AII,B48,CI,CII,CIII,E |
B100,CI,CII,CIII,E |
B100,CIII,E |
B100 |
AI,AII,CI,CII,CIII,D,E |
aSurface Components
bCore Lipids
VLDLy Found in Egg Yolk
"VLDLy" was coined to signify specific lipoproteins that
selectively deposit triacylglycerol to yolk follicles.
The average size of a VLDLy particle is 30nm, whereas a generic VLDL
particle is approximately 70nm.
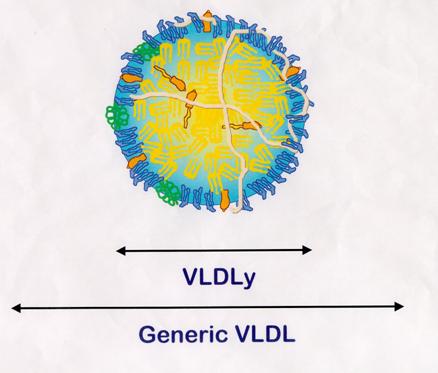
VLDLy
Metabolism
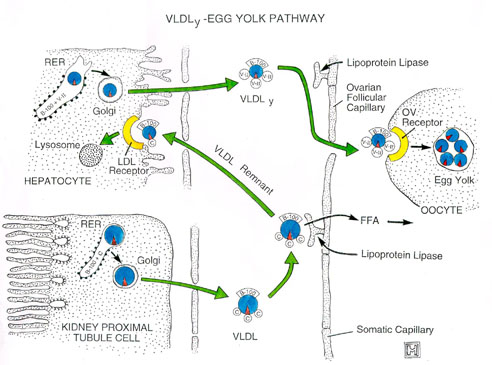
Theoretically, a 17g egg yolk that contains 2.8g of protein would contain
1.4g of apoB (49% total yolk protein, MW = 5.5 x 105).
Because VLDLy contains only one apoB protein per particle, this single egg yolk
would contain 1.5 x 1018 VLDLy particles. The hen would be
producing VLDLy particles at a rate of 1.5 x 1014 particles
per minute for seven days!!
Biochemistry of immune processes.
http://www.youtube.com/watch?v=Ys_V6FcYD5I&feature=related
Viruses, bacteria, fungi, and parasites
that enter the body of vertebrates of are recognized and attacked by the immune
system. Endogenous cells that have undergone alterations— e. g., tumor
cells—are also usually recognized as foreign and destroyed. The immune system
is supported by physiological changes in infected tissue, known as inflammation.
This reaction makes it easier for the immune cells to reach the site of
infection. Two different systems are involved in the immune response. The innate
immune system is based on receptors that can distinguish between bacterial
and viral surface structures or foreign proteins (known as antigens) and
those that are endogenous. With the help of these receptors, phagocytes bind
to the pathogens, absorb them by endocytosis, and break them down. The
complement system (see p. 298) is also part of the innate system. The acquired
(adaptive) immune system is based on the ability of the lymphocytes
to form highly specific antigen receptors “on suspicion,” without ever
having met the corresponding antigen. In humans, there are several billion
different lymphocytes, each of which carries a different antigen receptor. If
this type of receptor recognizes “its” cognate antigen, the lymphocyte carrying
it is activated and then plays its special role in the immune response. In
addition, a distinction is made between cellular and humoral immune responses.
The T lymphocytes (T cells)
are responsible for cellular immunity. They are named after the thymus,
in which the decisive steps in their differentiation take place. Depending on
their function, another distinction is made between cytotoxic T cells (green)
and helper T cells (blue).
http://www.youtube.com/watch?v=14koX2tbRzU&feature=related
http://www.youtube.com/watch?v=VOD5tuQ5wvo&feature=related
Humoral
immunity is based on the activity of the B lymphocytes (B
cells, light brown), which mature in the bone marrow. After activation by T
cells, B cells are able to release soluble forms of their specific antigen
receptors, known as antibodies (see p. 300), into the blood plasma. The
immune system’s “memory” is represented by memory cells. These are particularly
long–lived cells that can arise from any of the lymphocyte types described. Simplified
diagram of the immune response.
Pathogens
that have entered the body—e. g., viruses (top)—are taken up by antigen-presenting
cells (APCs) and proteolytically degraded (1). The viral fragments
produced in this way are then presented on the surfaces of these cells with the
help of special membrane proteins (MHC proteins; see p. 296) (2). The
APCs include B lymphocytes, macrophages, and dendritic cells such as the skin’s
Langerhans cells. The complexes of MHC proteins and viral fragments displayed
on the APCs are recognized by T cells that carry a receptor that matches the
antigen (“T-cell receptors”) (3). Binding leads to activation of the T
cell concerned and selective replication of it (4, “clonal selection”).
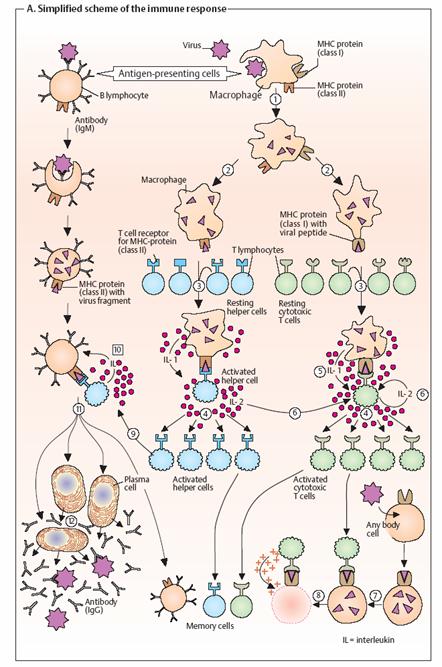
The proliferation of immune cells is stimulated by interleukins (IL).
These are a group of more than 20 signaling substances belonging to the
cytokine family (see p. 392), with the help of which immune cells communicate with
each other. For example, activated macrophages release IL-1 (5), while T
cells stimulate their own replication and that of other immune cells by
releasing IL-2 (6). Depending on their type, activated T cells have
different functions. Cytotoxic T cells (green) are able to recognize and
bind virusinfected body cells or tumor cells (7). They then drive the
infected cells into apoptosis (see p. 396) or kill them with perforin, a
protein that perforates the target cell’s plasma membrane (8). B
lymphocytes, which as APCs present viral fragments on their surfaces, are
recognized by helper T cells (blue) or their T cell receptors (9).
Stimulated by interleukins, selective clonal replication then takes place of B
cells that carry antigen receptors matching those of the pathogen (10).
Thesemature into plasma cells (11) and finally secrete large
amounts of soluble antibodies (12).
•
Antigen receptors
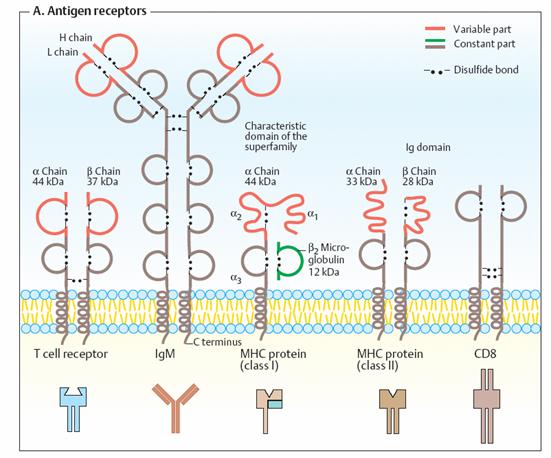
Many
antigen receptors belong to the immunoglobulin superfamily. The common
characteristic of these proteins is that they aremade up from “immunoglobulin
domains.” These are characteristically folded substructures consisting of
70–110 amino acids, which are also found in soluble immunoglobulins (Ig; see p.
300). The illustration shows schematically a few of the important proteins in
the Ig superfamily. They consist of constant regions (brown or green) and
variable regions (orange). Homologous domains are shown in the same colors in
each case. All of the receptors have transmembrane helices at the C terminus,
which anchor them to the membranes. Intramolecular and intermolecular disulfide
bonds are also usually found in proteins belonging to the Ig family. Immunoglobulin
M (IgM), a membrane protein on the surface of B lymphocytes, serves to bind
free antigens to the B cells. By contrast, T
cell receptors only bind antigens when they are presented by another cell
as a complex with an MHC protein (see below). Interaction between MHC-bound
antigens and T cell receptors is supported by co-receptors. This group
includes CD8, a membrane protein that is typical in cytotoxic T cells. T
helper cells use CD4 as a co-receptor instead (not shown). The
abbreviation “CD” stands for “cluster of differentiation.” It is the term for a
large group of proteins that are all located on the cell surface and can
therefore be identified by antibodies. In addition to CD4 and CD8, there are
many other co-receptors on immune cells
The MHC proteins are named after the “major histocompatibility
complex”—the DNA segment that codes for them. Human MHC proteins are also
known as HLA antigens (“human leukocyte-associated” antigens). Their
polymorphism is so large that it is unlikely that any two individuals carry the
same set of MHC proteins—except formonozygotic twins. Class I MHC
proteins occur in almost all nucleated cells. They mainly interact with
cytotoxic T cells and are the reason for the rejection of transplanted organs.
Class I MHC proteins are heterodimers (áâ). The â subunit is
also known as â2-microglobulin. Class II MHC proteins also consist
of two peptide chains, which are related to each other. MHC II molecules are
found on all antigen- presenting cells in the immune system. They serve for
interaction
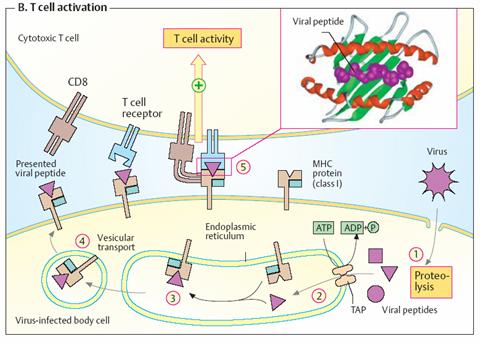
T-cell activation The
illustration shows an interaction between a virus-infected body cell (bottom)
and a CD8- carrying cytotoxic T lymphocyte (top). The infected cell breaks down
viral proteins in its cytoplasm (1) and transports the peptide fragments
into the endoplasmic reticulum with the help of a special transporter (TAP)
(2). Newly synthesized class I MHC proteins on the endoplasmic reticulum
are loaded with one of the peptides (3) and then transferred to the cell
surface by vesicular transport (4). The viral peptides are bound on the
surface of the á2 domain of the MHC protein in a depression formed by an
insertion as a “floor” and two helices as “walls” (see smaller illustration).
Supported by CD8 and other co-receptors, a T cell with a matching T cell
receptor binds to the MHC peptide complex (5). This binding activates
protein kinases in the interior of the T cell, which trigger a chain of
additional reactions (signal transduction). Finally, destruction of the
virus-infected cell by the cytotoxic T lymphocytes takes place.
Complement system
The complement system is part of the innate immune system (see p. 294).
It supports nonspecific defense against microorganisms. The system
consists of some 30 different proteins, the “complement factors,” which
are found in the blood and represent about 4% of all plasma proteins there.
When inflammatory reactions occur, the complement factors enter the infected
tissue and take effect there. The complement system works in three different
ways: Chemotaxis. Various complement factors attract immune cells that can
attack and phagocytose pathogens. Opsonization. Certain complement
factors (“opsonins”) bind to the pathogens and thereby mark them as targets for
phagocytosing cells (e. g., macrophages). Membrane attack. Other
complement factors are deposited in the bacterial membrane, where they create
pores that lyse the pathogen (see below).
•
The reactions that take place in the
complement system can be initiated in several ways. During the early phase of
infection, lipopolysaccharides and other structures on the surface of the
pathogens trigger the alternative pathway (right). If antibodies against
the pathogens become available later, the antigen– antibody complexes formed
activate the classic pathway (left). Acute-phase proteins are also able
to start the complement cascade (lectin pathway). Factors C1 to C4
(for “complement”) belong to the classic pathway, while factors B and
D form the reactive components of the alternative pathway. Factors C5
to C9 are responsible for membrane attack. Other components not
shown here regulate the system. As in blood coagulation (see p. 290), the early
components in the complement system are serine proteinases, which
mutually activate each other through limited proteolysis. They create a
self-reinforcing enzyme cascade.
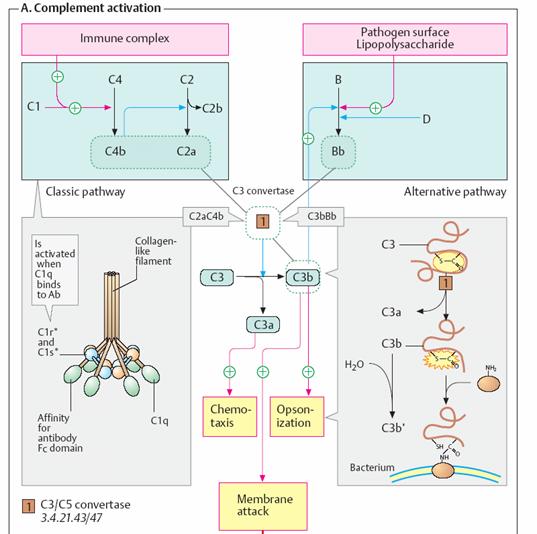
Factor C3, the products of which are involved in several
functions, is central to the complement system. The classic pathway is
triggered by the formation of factor C1 at IgG or IgM on the surface of
microorganisms (left). C1 is an 18-part molecular complex with three different
components (C1q, C1r, and C1s). C1q is shaped like a bunch of tulips, the
“flowers” of which bind to the Fc region of antibodies (left). This activates
C1r, a serine proteinase that initiates the cascade of the classic
pathway. First, C4 is proteolytically activated into C4b, which in turn cleaves
C2 into C2a and C2b. C4B and C2a together form C3 convertase [1], which
finally catalyzes the cleavage of C3 into C3a and C3b. Small amounts of C3b
also arise from non-enzymatic hydrolysis of C3.
The
classic pathway is triggered by the formation of factor C1 at IgG or IgM
on the surface of microorganisms (left). C1 is an 18-part molecular complex
with three different components (C1q, C1r, and C1s). C1q is shaped like a bunch
of tulips, the “flowers” of which bind to the Fc region of antibodies (left).
This activates C1r, a serine proteinase that initiates the cascade of
the classic pathway. First, C4 is proteolytically activated into C4b, which in
turn cleaves C2 into C2a and C2b. C4B and C2a together form C3 convertase [1],
which finally catalyzes the cleavage of C3 into C3a and C3b. Small amounts of
C3b also arise from non-enzymatic hydrolysis of C3. The alternative pathway starts
with the binding of factors C3b and B to bacterial lipopolysaccharides
(endotoxins). The formation of this complex allows cleavage of B by factor D,
giving rise to a second form of C3 convertase (C3bBb). Proteolytic
cleavage of factor C3 provides two components with different effects.
The reaction exposes a highly reactive thioester group in C3b, which
reacts with hydroxyl or amino groups. This allows C3b to bind covalently to
molecules on the bacterial surface (opsonization, right). In addition,
C3b initiates a chain of reactions leading to the formation of the membrane
attack complex Together with C4a and C5a (see below), the smaller product
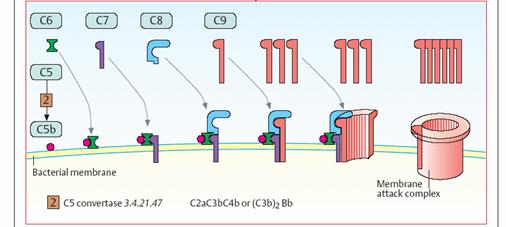
C3a promotes the inflammatory reaction and has chemotactic effects. The “late”
factors C5 to C9 are responsible for the development of the membrane attack
complex (bottom). They create an ion-permeable pore in the bacterial
membrane, which leads to lysis of the pathogen. This reaction is triggered by C5
convertase [2]. Depending on the type of complement activation, this enzyme
has the structure C4b2a3b or C3bBb3b, and it cleaves C5 into C5a
and C5b. The complex of C5b and C6 allows deposition of C7 in the bacterial
membrane. C8 and numerous C9 molecules—which form the actual pore—then bind to
this core. Antibodies
http://www.youtube.com/watch?v=lrYlZJiuf18
http://www.youtube.com/watch?v=Ys_V6FcYD5I&feature=related
•
Soluble antigen receptors, which are
formed by activated B cells (plasma cells; see p. 294) and released into the
blood, are known as antibodies. They are also members of the
immunoglobulin family (Ig; see p. 296). Antibodies are an important part of the
humoral immune defense system. They have no antimicrobial properties
themselves, but support the cellular immune system in various ways: 1. They
bind to antigens on the surface of pathogens and thereby prevent them from
interacting with body cells (neutralization; see p. 404, for example).
2. They link single-celled pathogens into aggregates (immune complexes), which
are more easily taken up by phagocytes (agglutination). 3. They activate
the complement system (see p. 298) and thereby promote the innate immune
defense system (opsonization). In addition, antibodies have become
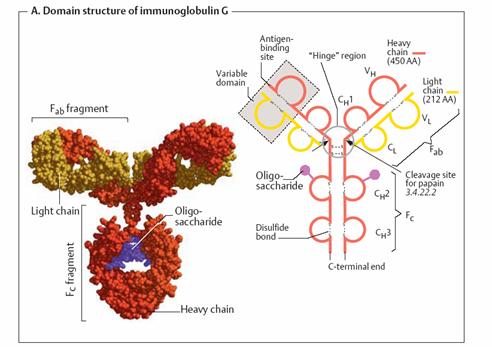
indispensable aids in medical and biological diagnosis. Domain structure of
immunoglobulin G _
Type G
immunoglobulins (IgG) are quantitatively the most important antibodies
in the blood,where they form the fraction of ã-globulins (see p. 276).
IgGs (mass 150 kDa) are tetramers with two heavy chains (H chains; red
or orange) and two light chains (L chains; yellow). Both H chains are
glycosylated (violet; see also p. 43). The proteinase papain cleaves IgG
into two Fab fragments and one Fc fragment. The Fab (“antigen-binding”)
fragments, which each consist of one L chain and the N-terminal part of an H
chain, are able to bind antigens. The Fc (“crystallizable”) fragment is
made up of the C-terminal halves of the two H chains. This segment serves to
bind IgG to cell surfaces, for interaction with the complement system and
antibody transport. Immunoglobulins are constructed in a modular fashion from
several immunoglobulin domains (shown in the diagram on the right in Ω
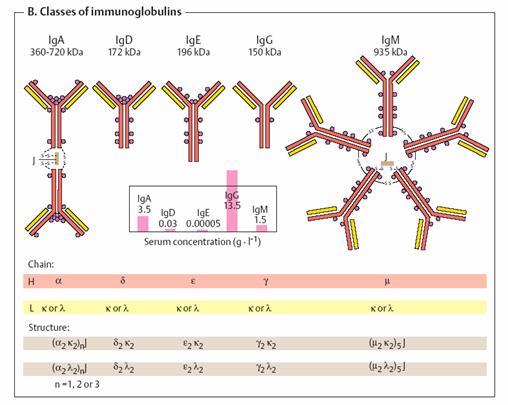
form). Classes of immunoglobulins _
http://www.youtube.com/watch?v=mUXIK5gGD1k
Human
immunoglobulins are divided into five classes. IgA (with two subgroups),
IgD, IgE, IgG (with four subgroups), and IgM are defined by their
H chains, which are designated by the Greek letters á, ä, å,
ã, and µ. By contrast, there are only two types of L chain (ê
and ë). IgD and IgE (like IgG) are tetramers with the structure H2L2. By
contrast, soluble IgA and IgM are multimers that are held together by disulfide
bonds and additional J peptides (joining peptides). The antibodies have
different tasks. IgMs are the first immunoglobulins formed after contact
with a foreign antigen. Their early forms are located on the surface of B cells
(see p. 296), while the later forms are secreted from plasma cells as
pentamers. Their action targets microorganisms in particular. Quantitatively, IgGs
are the most important immunoglobulins (see the table showing serum
concentrations). They occur in the blood and interstitial fluid. As they can
pass the placenta with the help of receptors, they can be transferred from
mother to fetus. IgAs mainly occur in the intestinal tract and in body
secretions. IgEs are found in low concentrations in the blood. As they
can trigger degranulation of mast cells (see p. 380), they play an important
role in allergic reactions. The function of IgDs is still unexplained.
Their plasma concentration is also very low.
Causes of antibody variety _
There are three reasons for the extremely wide variability of
antibodies: 1. Multiple genes. Various genes are available to code for
the variable protein domains. Only one gene from among these is selected and
expressed. 2. Somatic recombination. The genes are divided into several
segments, of which there are various versions. Various (“untidy”) combinations
of the segments during lymphocyte maturation give rise to randomly combined new
genes (“mosaic genes”). 3. Somaticmutation. During differentiation of B
cells into plasma cells, the coding genes mutate. In this way, the “primordial”
germline genes can become different somatic genes in the
individual B cell clones.
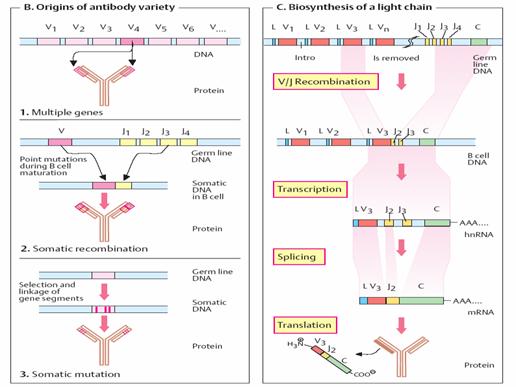
Biosynthesis of a light chain _
We can look at the basic features of the genetic organization and
synthesis of immunoglobulins using the biosynthesis of a mouse ê chain as
an example. The gene segments for this light chain are designated L, V, J, and
C. They are located on chromosome
The V segments, of which there are 150 different variants, code
formost of the variable domains (95 of the 108 amino acids). L and V segments
always occur in pairs—in tandem, so to speak. By contrast, there are only five
variants of the J segments (joining segments) at most. These code for a
peptide with 13 amino acids that links the variable part of the ê chains
to the constant part. A single C segment codes for the constant part of
the light chain (84 amino acids). During the differentiation of B lymphocytes,
individual V/J combinations arise in each B cell. One of the 150 L/V
tandem segments is selected and linked to one of the five J segments.
The immune system is a complex, dynamic, and beautifully orchestrated
mechanism with enormous responsibility. It defends against foreign invasion by
microorganisms, screens out cancer cells, adapts as we grow, and modifies how
we interact with our environment. When it malfunctions, disease, cancer or
death can occur. Although it is not necessary to understand all the intimate
details of the immune system, it is wise to have a basic grasp of its
functions. More precisely, we should understand how to stay healthy.
Training the immune system -- the
"J" curve
It appears that the immune system has a training effect, similar to
other areas of physiology (e.g., cardiovascular, muscular). In other words, a
balanced training program of exercise and rest leads to better performance.
Studies in the laboratory and epidemiological observations have shown improved
immune function and fewer URIs in athletes as compared to their couch-potato
counterparts. This is especially true in older athletes and it appears that
regular exercise can help attenuate the age related decline in immune function.
On the other hand, too much exercise can lead to a dramatically
increased risk of URIs. The stress of strenuous exercise transiently suppresses
immune function. This interruption of otherwise vigorous surveillance can
provide an "open window" for a variety of infectious diseases --
notably viral illnesses -- to take hold. This is especially true following
single bouts of excessive exercise. For example, it has been observed that
two-thirds of participants developed URIs shortly after completing an
ultramarathon. Similarly, cumulative overtraining weakens the athlete's immune
system, leading to frequent illness and injury.
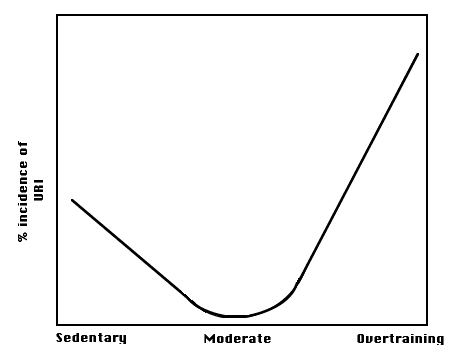
The best model that accommodates clinical observations and laboratory
experiments is described by the "J"-curve ( Fig. 1). It is important
to note that this curve is individualized. What is moderate training for some
is overtraining for others.
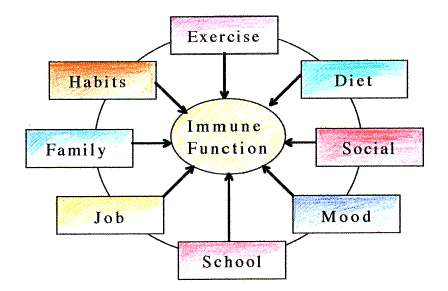
Stress is cumulative
In
addition to strenuous exercise, other forms of stress may also transiently
suppress immune function. Since exercise is not the only stress factor, an
athlete must consider a host of other variables. There are job
responsibilities, family obligations, social interactions, financial concerns
and other components that shape our lives. The sum of all of these affects a
central axis in the body which ultimately influences immune function. Some of
these (e.g., exercise) are under our direct control, and others only partially
or not at all. Recognizing when excess stress occurs is easier if it just comes
from one source. However, all too often it is the sum of many small, difficult to
recognize changes that tips the scales and sends the athlete into the whirlpool
of overtraining and immunosuppression. Alone and in isolation these small
changes would be manageable, but combined they can overwhelm. (Fig. 2.)
Recommendations
Currently,
the best way to stay healthy is to listen to your body. Recognizing the early
warning signs and adapting the training schedule accordingly can help keep you
healthy. In that light, here are some points to ponder and a few
recommendations,
·
Keep a training log. In addition to
recording workouts, keep a fatigue score (scale 0-5). It is expected that a
hard workout will make you tired, so it is more important to note the
cumulative "feel" during the day. Granted, the scale is
individualized and subjective, but this simple tool is very useful. If you
notice that your fatigue is progressively increasing over days or weeks, then
it is time to add more rest.
·
A properly constructed training
program that allows for rest and recovery will help head off problems before
they start. Periodization
is a way to achieve that goal.
·
Record your resting morning heart
rate. A progressive increase may tip you off that you are exceeding your
ability to recover.
·
Anticipate added stress in advance
(e.g. new job) and adjust the workout schedule correspondingly. A small amount
of rest early will prevent a bigger problem later.
·
To make sure your anti-oxidant
defense system is tuned up, eat five servings of fruit or vegetables per day.
Note: vitamin supplements do not appear to have the same benefits as fruits and
vegetables.
·
Heed your body's early warning signs,
o
Disordered sleep (too much or
insomnia)
o
Loss of interest in pleasurable
activities
o
Moodiness
or depression
o
Excessive
muscle soreness
o
Poor concentration. Lack of mental energy.
o
Altered
appetite.
o
Frequent
injury or illness
o
Lack
of physical energy
·
Get an annual influenza vaccine
(usually available each year starting in October)
·
Because frequent URIs or unrelenting
fatigue may be a sign of an underlying illness, it is recommended that you
consult your physician.
The Anatomy of the Immune System
The organs of the immune system are stationed
throughout the body. They are generally referred to as lymphoid organs because
they are concerned with the growth, development, and deployment of lymphocytes,
the white cells that are the key operatives of the immune system. Lymphoid
organs include the bone marrow and the thymus, as well as lymph nodes, spleen,
tonsils and adenoids, the appendix, and clumps of lymphoid tissue in the small
intestine known as Peyer's patches. The blood and lymphatic vessels that carry
lymphocytes to and from the other structures can also be considered lymphoid
organs.
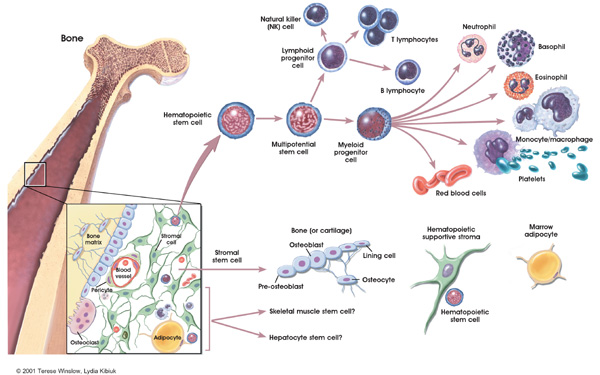
Cells destined to become immune cells, like all other
blood cells, are produced in the bone marrow, the soft tissue in the hollow
shafts of long bones. The descendants of some so-called stem cells become
lymphocytes, while others develop into a second major group of immune cells
typified by the large, cell-and particle-devouring white cells known as
phagocytes.
The two major classes of lymphocytes are B cells and T
cells. B cells complete their maturation in the bone marrow. T cells, on the
other hand, migrate to the thymus, a multilobed organ that lies high behind the
breastbone. There they multiply and mature into cells capable of producing
immune response-that is, they become immunocompetent. In a process referred to
as T cell "education," T cells in the thymus learn to distinguish
self cells from nonself cells; T cells that would react against self antigens
are eliminated.
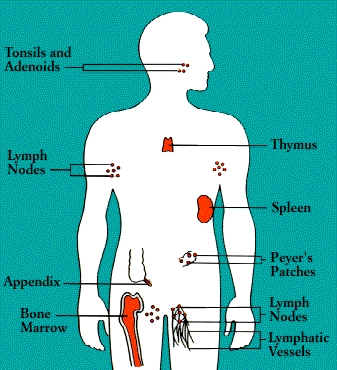
Upon exiting the bone marrow and thymus, some
lymphocytes congregate in immune organs or lymph nodes. Others-both B and T
cells-travel widely and continuously throughout the body. They use the blood circulation
as well as a bodywide network of lymphatic vessels similar to blood vessels.
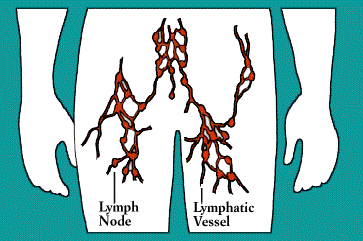
Laced along the lymphatic routes-with clusters in the
neck, armpits, abdomen, and groin-are small, bean-shaped lymph nodes. Each
lymph node contains specialized compartments that house platoons of B
lymphocytes, T lymphocytes, and other cells capable of enmeshing antigen and
presenting it to T cells. Thus, the lymph node brings together the several
components needed to spark an immune response.
The spleen, too, provides a meeting ground for immune
defenses. A fist-sized organ at the upper left of the abdomen, the spleen
contains two main types of tissue: the red pulp that disposes of worn-out blood
cells and the white pulp that contains lymphoid tissue. Like the lymph nodes,
the spleen's lymphoid tissue is subdivided into compartments that specialize in
different kinds of immune cells. Microorganisms carried by the blood into the
red pulp become trapped by the immune cells known as macrophages. (Although
people can live without a spleen, persons whose spleens have been damaged by
trauma or by disease such as sickle cell anemia, are highly susceptible to
infection; surgical removal of the spleen is especially dangerous for young
children and the immunosuppressed.)
Nonencapsulated clusters of lymphoid tissue are found in many parts of
the body. They are common around the mucous membranes lining the respiratory
and digestive tracts-areas that serve as gateways to the body. They include the
tonsils and adenoids, the appendix, and Peyer's patches.
The lymphatic vessels carry lymph, a clear fluid that bathes the body's
tissues. Lymph, along with the many cells and particles it carries-notably
lymphocytes, macrophages, and foreign antigens, drains out of tissues and seeps
across the thin walls of tiny lymphatic vessels. The vessels transport the mix
to lymph nodes, where antigens can be filtered out and presented to immune
cells.
Additional lymphocytes reach the lymph nodes (and other immune tissues)
through the bloodstream. Each node is supplied by an artery and a vein;
lymphocytes enter the node by traversing the walls of the very small
specialized veins.
All lymphocytes exit lymph nodes in lymph via outgoing lymphatic
vessels. Much as small creeks and streams empty into larger rivers, the
lymphatics feed into larger and larger channels. At the base of the neck, large
lymphatic vessels merge into the thoracic duct, which empties its contents into
the bloodstream.
Once in the bloodstream, the lymphocytes and other assorted immune cells
are transported to tissues throughout the body. They patrol everywhere for
foreign antigens, then gradually drift back into the lymphatic vessels, to
begin the cycle all over again
Disorders of the Immune System: Allergy
http://www.youtube.com/watch?v=NFTL51FvX4Q&feature=related
The most
common types of allergic reactions-hay fever, some kinds of asthma, and
hives-are produced when the immune system response to a false alarm. In a
susceptible person, a normally harmless substance-grass pollen or house dust,
for example-is perceived as a threat and is attacked.
Such allergic
reactions are related to the antibody known as immunoglobulin E. Like other antibodies,
each IgE antibody is specific; one reacts against oak pollen, another against
ragweed. The role of IgE in the natural order is not known, although some
scientists suspect that it developed as a defense against infection by
parasitic worms.
The
first time an allergy-prone person is exposed to an allergen, he or she makes
large amounts of the corresponding IgE antibody. These IgE molecules attach to
the surfaces of mast cells (in tissue) or basophils (in the circulation). Mast
cells are plentiful in the lungs, skin, tongue, and linings of the nose and
intestinal tract.
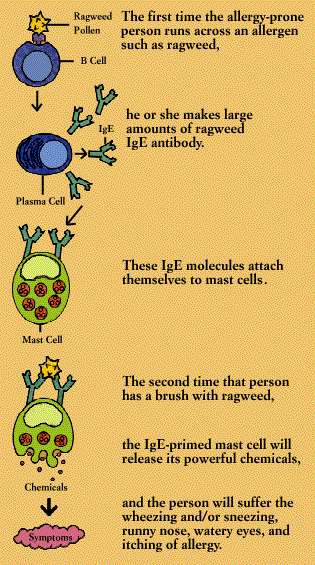
When
an IgE antibody siting on a mast cell or basophil encounters its specific
allergen, the IgE antibody signals the mast cell or basophil to release the
powerful chemicals stored within its granules. These chemicals include
histamine, heparin, and substances that activate blood platelets and attract
secondary cells such as eosinophils and neutrophils. The activated mast cell or
basophil also synthesizes new mediators, including prostaglandins and
leukotrienes, on the spot.
It
is such chemical mediators that cause the symptoms of allergy, including
wheezing, sneezing, runny eyes and itching. They can also produce anaphylactic
shock, a life-threatening allergic reaction characterized by swelling of body
tissues, including the throat, and a sudden fall in blood pressure.
Autoimmune
Diseases
Sometimes the
immune system's recognition apparatus breaks down, and the body begins to
manufacture antibodies and T cells directed against the body's own
constituents-cells, cell components, or specific organs. Such antibodies are
known as autoantibodies, and the diseases they produce are called autoimmune
diseases. (Not all autoantibodies are harmful; some types appear to be integral
to the immune system's regulatory scheme.)
Autoimmune
reactions contribute to many enigmatic diseases. For instance, autoantibodies
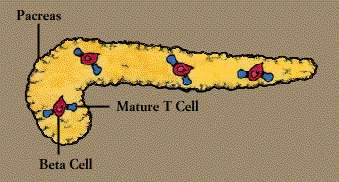
to red blood cells can cause anemia, autoantibodies to pancreas cells
contribute to juvenile diabetes, and autoantibodies to nerve and muscle cells
are found in patients with the chronic muscle weakness known as myasthenia
gravis. Autoantibody known as rheumatoid factor is common in persons with
rheumatoid arthritis.
Persons with systemic
lupus erythematosus (SLE), whose symptoms encompass many systems, have
antibodies to many types of cells and cellular components. These include
antibodies directed against substances found in the cell's nucleus-DNA, RNA, or
proteins-which are known as antinuclear antibodies, or ANAs. These antibodies
can cause serious damage when they link up with self antigens to form
circulating immune complexes, which become lodged in body tissue and set off
inflammatory reactions (Immune Complex Diseases).
Autoimmune
diseases affect the immune system at several levels. In patients with SLE, for
instance, B cells are hyperactive while suppressor cells are underactive; it is
not clear which defect comes first. Moreover, production of IL-2 is low, while
levels of gamma interferon are high. Patients with rheumatoid arthritis, who
have a defective suppressor T cell system, continue to make antibodies to a
common virus, whereas the response normally shuts down after about a dozen
days.
No one knows
just what causes an autoimmune disease, but several factors are likely to be
involved. These may include viruses and environmental factors such as exposure
to sunlight, certain chemicals, and some drugs, all of which may damage or
alter body cells so that they are no longer recognizable as self. Sex hormones
may be important, too, since most autoimmune diseases are far more common in
women than in men.
Heredity also
appears to play a role. Autoimmune reactions, like many other immune responses,
are influenced by the genes of the MHC. A high proportion of human patients
with autoimmune disease have particular histocompatibility types. For example,
many persons with rheumatoid arthritis display the self marker known as
HLA-DR4.
Many types of
therapies are being used to combat autoimmune diseases. These include
corticosteroids, immunosuppressive drugs developed as anticancer agents,
radiation of the lymph nodes, and plasmapheresis, a sort of "blood
washing" that removes diseased cells and harmful molecules from the circulation.
Immune
Complex Diseases
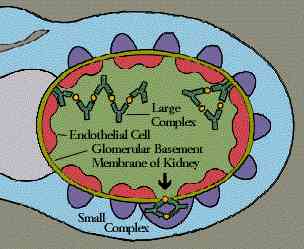
Immune
complexes are clusters of interlocking antigens and antibodies. Under normal
conditions immune complexes are rapidly removed from the bloodstream by
macrophages in the spleen and Kupffer cells in the liver. In some circumstances,
however, immune complexes continue to circulate. Eventually they become trapped
in the tissues of the kidneys, lung, skin, joints, or blood vessels. Just where
they end up probably depends on the nature of the antigen, the class of
antibody-IgG, for instance, instead of IgM-and the size of the complex. There
they set off reactions that lead to inflammation and tissue damage.
Immune
complexes work their damage in many diseases. Sometimes, as is the case with
malaria and viral hepatitis, they reflect persistent low-grade infections.
Sometimes they arise in response to environmental antigens such as the moldy
hay that causes the disease known as farmer's lung. Frequently, immune
complexes develop in autoimmune disease, where the continuous production of
autoantibodies overloads the immune complex removal system.
Immunodeficiency
Diseases
Lack of one
or more components of the immune system results in immunodeficiency disorders.
These can be inherited, acquired through infection or other illness, or produced
as an inadvertent side effect of certain drug treatments.
People with
advanced cancer may experience immune deficiencies as a result of the disease
process or from extensive anticancer therapy. Transient immune deficiencies can
develop in the wake of common viral infections, including influenza, infectious
mononucleosis, and measles. Immune responsiveness can also be depressed by
blood transfusions, surgery malnutrition, and stress.
Some children
are born with defects in their immune systems. Those with flaws in the B cell
components are unable to produce antibodies (immunoglobulins). These
conditions, known as agammaglobulinemias or hypogammaglobulinemias, leave the
children vulnerable to infectious organisms; such disorders can be combated
with injections of immunoglobulins.
Other
children, whose thymus is either missing or small and abnormal, lack T cells.
The resultant disorders have been treated with thymic transplants.
Very rarely,
infants are born lacking all the major immune defenses; this is known as severe
combined immunodeficiency disease (SCID). Some children with SCID have lived
for years in germ-free rooms and "bubbles." A few SCID patients have
been successfully treated with transplants of bone marrow (Bone Marrow
Transplants).
The devastating
immunodeficiency disorder known as the acquired immunodeficiency syndrome
(AIDS) was first recognized in 1981. Caused by a virus (the human
immunodeficiency virus, or HIV) that destroys T4 cells and that is harbored in
macrophages as well as T4 cells, AIDS is characterized by a variety of unusual
infections and otherwise rare cancers. The AIDS virus also damages tissue of
the brain and spinal cord, producing progressive dementia.
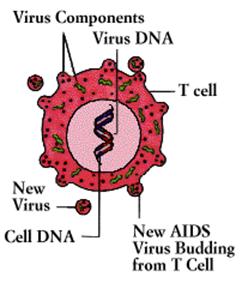
AIDS
infections are known as "opportunistic" because they are produced by
commonplace organisms that do not trouble people whose immune systems are
healthy, but which take advantage of the "opportunity" provided by an
immune defense in disarray. The most common infection is an unusual and
life-threatening form of pneumonia caused by a one-celled organism (a Protozoa)
called Pneumocystis carinii. AIDS patients are also susceptible to unusual
lymphomas and Kaposi's sarcoma, a rare cancer that results from the abnormal
proliferation of endothelial cells in the blood vessels.
Some
persons infected with the AIDS virus develop a condition known as AIDS-related
complex, or ARC, characterized by fatigue, fever, weight loss, diarrhea, and
swollen lymph glands. Yet other persons who are infected with the AIDS virus
apparently remain well; however, even though they develop no symptoms, they can
transmit the virus to others.
AIDS
is a contagious disease, spread by intimate sexual contact, by direct
inoculation of the virus into the bloodstream, or from mother to child during
pregnancy. Most of the AIDS cases in the United States have been found among
homosexual and bisexual men with multiple sex partners, and among intravenous
drug abusers. Others have involved men who received untreated blood products
for hemophilia; persons who received transfusions of inadvertently contaminated
blood-primarily before the AIDS virus was discovered and virtually eliminated
from the nation's blood supply with a screening test; the heterosexual partners
of persons with AIDS; and children born to infected mothers.
There
is presently no cure for AIDS, although the antiviral agent zidovuzine (AZT)
appears to hold the virus in check, at least for a time. Many other
antiretroviral drugs are being tested, as are agents to bolster the immune
system and agents to prevent or treat opportunistic infections. Research on
vaccines to prevent the spread of AIDS is also under way.
Cancers of
the Immune System
Cells of the
immune system, like those of other body systems, can proliferate uncontrollably;
the result is cancer. Leukemias are caused by the proliferation of white blood
cells, or leukocytes. The uncontrolled growth of antibody-producing (plasma)
cells can lead to multiple myeloma. Cancers of the lymphoid organs, known as
lymphomas, include Hodgkin's disease. These disorders can be treated-some of
them very successfully-by drugs and/or irradiation.
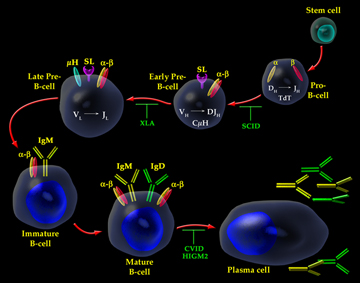
The Human Immune Response
System
An overview of the system
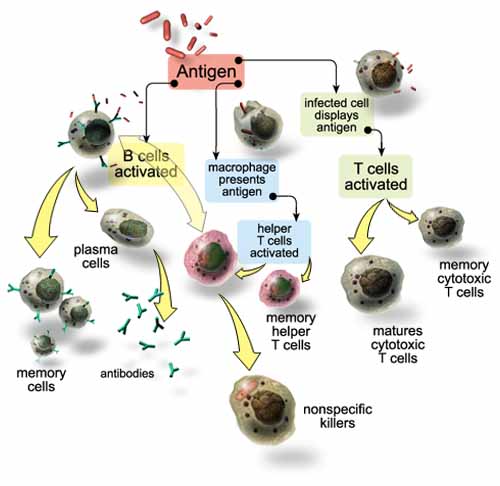
An overview of the system
The human immune response system recognizes pathogens and acts to remove,
immobilize, or neutralize them. The immune system is antigen-specific
(responding to specific molecules on a pathogen) and has memory (its defense to
a pathogen is encoded for future activation). The immune system relies on
several components to fight an infecting pathogen. T cells are lymphocytes that
circulate between the blood, lymph, and lymphoid organs to trigger a systemic
immune response with antigen-receptors on the T cell membrane. B cells are
lymphocytes that activate the primary immune response when antigens bind to
their receptors, causing the B cells to proliferate. Daughter cells of B cells
later differentiate into antibody-releasing plasma cells. B cells also comprise
the immune system's memory (see diagram).
Antibodies, also called immunoglobulins, are divided into five classes by
structure and function, enabling them to recognize a wide spectrum of antigens.
Antibody functions include complement fixation that can lead to antigen-cell
lysis (rupture) and can cause inflammation. Antibodies also generate a
neutralization response where viruses and bacteria are destroyed by phagocytes.
Agglutination, or clumping together, of foreign cells are caused by B cells'
promotion of complex cross-linking of antibodies binding to antigens. These
agglutinated cells are phagocytized. B cells are cloned in massive quantities
for a single specific antigen.
The
human immune response to T. cruzi infection is inadequate; it provides
only a partial defense at best. The immune system's response at its worst
causes the defense mechanisms to turn on the body it is intended to protect,
thus often causing more harm to the person than does T. cruzi.

As T. cruzi immunizes humans to their own antigens, human
antibodies attack myocardial and neural cells.
Complement in humans does not become activated solely by T. cruzi
invasion; antibodies must be present for complement to bind to a specific T.
cruzi antigen. This allows T. cruzi to have time to infect human
tissue. Parasite strain and an individual's immune competence are prime factors
in determining the T. cruzi's pathology of an individual.
Once infected with T. cruzi, humans acquire partial immunity or
resistance to the severe pathologies of Chagas' disease's acute phase through
subsequent infections of T. cruzi. This guards many individuals who live
in highly endemic areas from the acute symptoms of chagas. Complete removable
of the parasite from these individuals would risk the onset of acute chagas
through future infection, which is deadly - especially for children.
T. cruzi
incorporates certain host cell membrane proteins onto its surface thereby
masking its antigenic signal to the immune system's lymphocytes. T. cruzi
can also cleave antibody molecules on its surface thereby escaping the immune
response's detection. T. cruzi frequently invade monocytes, a
circulating phagocyte. Intracellular phagocytosis bring amastigotic T. cruzi
into tissue cells where they can proliferate. Once inside tissue cells, T.
cruzi are undetected by immune response. Trypomastigotes remain in the
blood stream for a short period of time so that the T. cruzi-specific
immunoglobulins don't have sufficient time to be activated. T. cruzi
employs successful strategies to escape the remarkably potent immune response
system. By masking themselves or by eluding the response mechanisms, the
parasite is able to adapt to survive and continue the life of the species.
Immune response that
damages the human body
Unintentional
damage is done to the
body's otherwise healthy tissue as the response system attacks what it
recognizes as a trigger for a defensive response but does not recognize that it
is attacking itself. This is what's known as an autoimmune reaction.
Autoimmune
responses are responsible in large part for the destructive symptoms of Chagas
disease. This pathology is referred to as immunopathology. Severe inflammation
occurs around tissue that embody amastigotes as the amastigotes release
themselves from the tissue's dead cells. Among the tissue most often encysted
is myocardial neural plexes. Plexes are networks of nerves that serve a variety
of organs and functions. Digestive system neural plexes are targets as well,
namely in the colon and esophagus. During the acute phase of chagas, B and T
cells are incited to produce antibodies. Since T. cruzi is able to mask
its presence in the blood, these antibodies do not attack T. cruzi but
instead go after cell membrane antigenic components called epitopes, that the
body's healthy cells and T. cruzi share. Research is being done to
isolate the epitope and how T. cruzi uses it to elude recognition by the
immune system.
Scientists work to find a cure to T. cruzi's
infecting the human species. As research continues into how T. cruzi
uses the human body as a host, the disciplines of parasitology and immunology
learn much about how these organisms adapt and thrive in changing environments.
T. cruzi proves to be a formidable opponent in the fight.
Mediated by Macrophages
Mediated by Lymphocytes and mast
cells
Identical in all Individuals Depends on
Exposure to Pathogens
Fixed by Evolution
Evolving within individuals
Parasites, worms, chemicals, toxins generic viruses,
bacteria lipoproteins, lipocarbohydrates bacterial
DNA
Phagocytosis
(engulfment)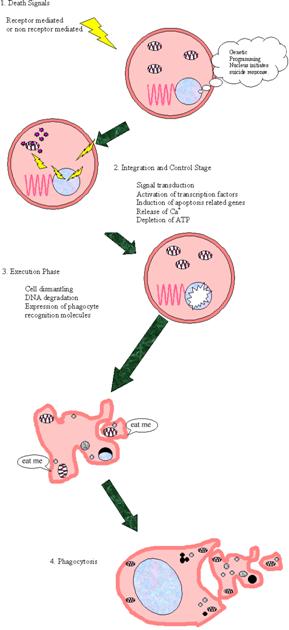
Activation of Antibodies
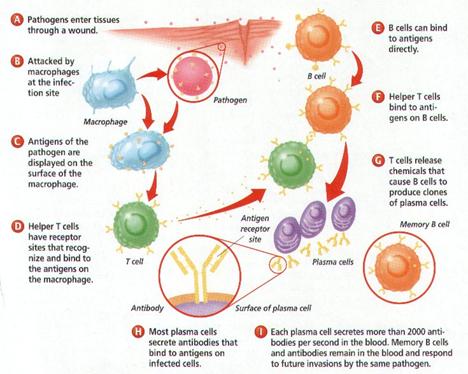
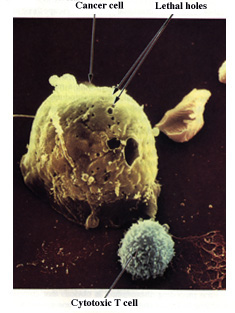
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|