Investigation of fat soluble vitamins functional role in metabolism and cell functions realization.
Fat-soluble vitamins
Although fat-soluble vitamins have been studied
intensively and widely used in human nutrition, we know less about their specific
biological function than about the water-soluble vitamins.
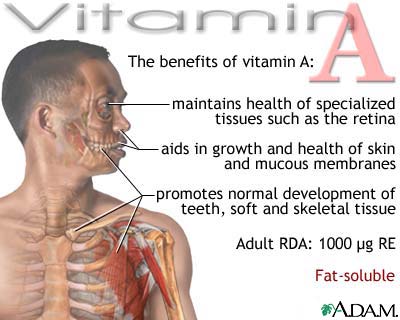
Vitamin A.
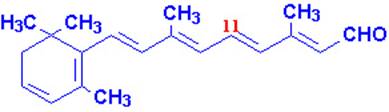
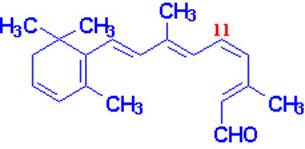
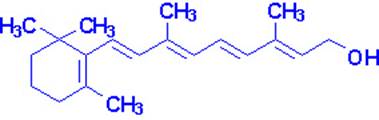
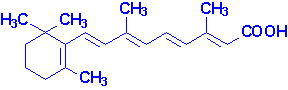
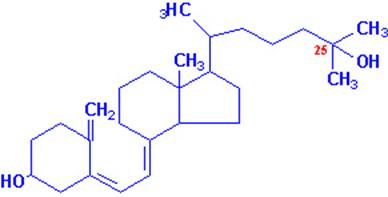
Vitamin A occurs in two common forms, vitamin A1, or retinol, the form most common in
mammalian tissues and marine fishes, and vitamin, A2, common in freshwater
fishes. Both are isoprenoid compounds containing a
six-membered carbocyclic
ring and an eleven-carbon side chain.
http://www.youtube.com/watch?v=dcw1m31zuTE
Vitamin A
Vitamin A consists of three biologically active
molecules, retinol, retinal (retinaldehyde) and retinoic acid.
|
|
|
|
All-trans-retinal |
11-cis-retinal |
|
|
|
|
Retinol |
Retinoic Acid |
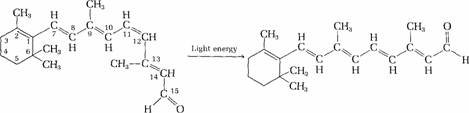
Carotenoids are provitamins
of vitamin A. Carotenoids widely distributed in
plants, particularly a-, b-, and g-carotene. The carotenes have no vitamin A
activity but are converted into vitamin A by enzymatic reactions in the
intestinal mucosa and the liver. b-Carotene, a symmetrical molecule, is cleaved
in its center to yield two molecules of retinol. Retinol occurs in the tissues
of mammals and is transported in the blood.
In vitamin A deficiency young
persons fail to grow, the bones and nervous system fail to develop properly,
the skin becomes dry and thickened, the kidneys and various glands degenerate,
and both males and females become sterile.
Although all tissues appear
to be disturbed by vitamin A deficiency, the eyes are most conspicuously
affected. In infants and young children the condition known as xerophthalmia
("dry eyes") is an early symptom of deficiency and is a common cause
of blindness in some tropical areas where nutrition is generally poor. In
adults an early sign of vitamin A deficiency is nightblindness, a deficiency in
dark adaptation, which is often used as a diagnostic test.
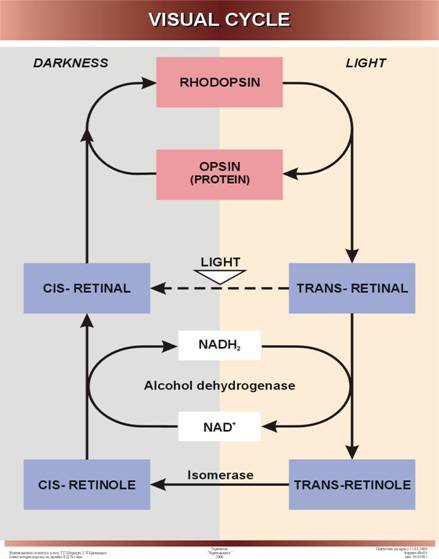
Detailed information is
available on the role of vitamin A in the visual_cycle in vertebrates. The
human retina contains two types of light-sensitive photoreceptor cells. Rod-cells are adapted to sensing low
light intensities, but not colors; they are the cells involved in night vision,
whose function is impaired by vitamin A deficiency. Cone cells, which sense colors, are adapted for high light
intensities.
Retinal rod cells contain many membrane
vesicles that serve as light receptors. About one-half of the protein in the
membrane of these vesicles consists of the light-absorbing protein rhodopsin (visual purple). Rhodopsin
consists of a protein, opsin,
and tightly bound 11-cis-retinal, the
aldehyde of vitamin A. When rhodopsin
is exposed to light, the bound 11-cis-retinal undergoes transformation into
all-trans-retinal, which causes a substantial change in the configuration of
the retinal molecule. This reaction is nonenzymatic.
The isomerization of retinal is followed by a series
of other molecular changes, ending in the dissociation of the rhodopsin to yield free opsin and
all-trans-retinal, which functions as a trigger setting off the nerve impulse.
11-cis-retinal all-trans-retinal
In order for rhodopsin
to be regenerated from opsin and all-trans-retinal,
the latter must undergo isomerization back to
11-cis-retinal. This appears to occur in a sequence of enzymatic reactions
catalyzed by two enzymes:
retinal-reductase
all-trans-retinal
+ NADH + H+ → all-trans-retinol + NAD+
retinol-isomerase
all-trans-retinol →
11-cis-retinol
retinal-reductase
11-cis-retinol +
NAD+ → 11-cis-retinal + NADH + H+
The 11-cis-retinal so formed now recombines with opsin to yield rhodopsin, thus
completing the visual cycle.
Since vitamin A deficiency affects all tissues
of mammals, not the retina alone, the role of retinal in the visual cycle does
not represent the entire action of vitamin A. It appears possible that vitamin
A may play a general role in:
- the transport of Ca2+ across
certain membranes; such a more general role might explain the effects of
vitamin A deficiency and excess on bony and connective tisues;
-
processes of growth and cell
differentiation;
-
processes of glycoproteins formation whoch are
the components of the biological mucosa .
The vitamin A requirement of man - 1,5-2
milligram per day.
Vitamin A is met in large part by green and
yellow vegetables, such as lettuce, spinach, sweet potatoes, and carrots, which
are rich in carotenes. Fish-liver oils are particularly rich in vitamin A.
However, excessive intake of vitamin A is toxic and leads to easily fractured,
fragile bones in children, as well as abnormal development of the fetus.
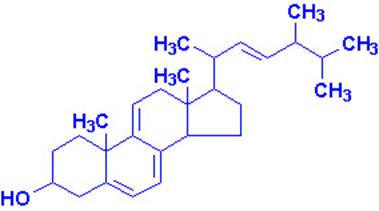
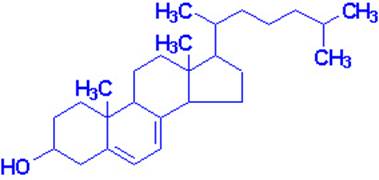
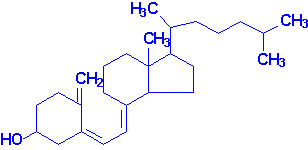
Vitamin D
Most important are vitamin D2, or ergocalciferol,
and vitamin D3, or cholecalciferol, the form normally found in mammals.
These compounds may be regarded as steroids.
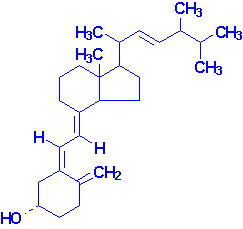
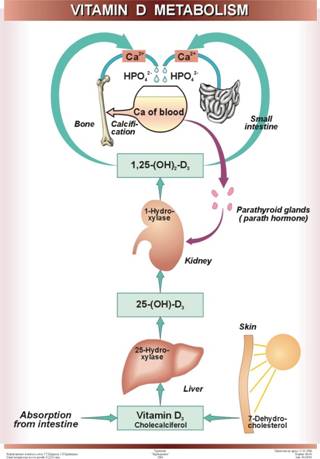
It is now known that 7-dehydrocholesterol in the skin is the natural precursor of cholecalciferol in man; the conversion requires irradiation
of the skin by sunlight. On a normal unsupplemented
diet this is the major route by which people usually acquire vitamin D.
Vitamin D is a
steroid hormone that functions to regulate specific gene expression following
interaction with its intracellular receptor. The biologically active form of
the hormone is 1,25-dihydroxy vitamin D3 (1,25-(OH)2D3,
also termed calcitriol). Calcitriol functions primarily to regulate calcium and
phosphorous homeostasis.
http://www.youtube.com/watch?v=JwPVibQ6_3Y&feature=related
http://www.youtube.com/watch?v=onSPZ0aBUKM&feature=related
http://www.youtube.com/watch?v=xwNhd2pQL0k&feature=related
|
|
|
|
Ergosterol |
Vitamin D2 |
|
|
|
|
7-Dehydrocholesterol |
Vitamin D3 |
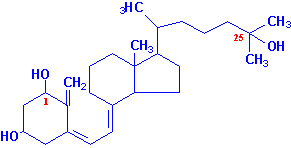
Cholecalciferol is converted into its derivative - 25-hydroxycholecalciferol. This product
is more active biologically than cholecalciferol and
it has been found to be the main circulating form of vitamin D in animals,
formed in the liver. But 25-hydroxycholecalciferol was found to be metabolized
further to 1,25-dihydroxycholecalciferol in kidneys. This compound is still
more active; its administration produces rapid stimulation of Ca2+
absorption by the intestine.
|
|
|
|
25-hydroxyvitamin D3 |
1,25-dihydroxyvitamin
D3 |
So the kidney is the site of formation of 1,25-dihydroxycholecalciferol,
which now appears to be the biologically active form of vitamin D, capable of
acting directly on its major targets, the small intestine and the bones.
1,25-dihydroxycholecalciferol promotes
absorption of Ca2+ from the intestine into the blood, through its
ability to stimulate the biosynthesis of specific protein(s) that participate
in transport or binding of Ca2+ in the intestinal mucosa. This role
of 1,25-dihydroxycholecalciferol is integrated with the action of parathyroid
hormone. Whenever the Ca2+ concentration of the blood becomes
lower than normal, the parathyroid glands secrete larger amounts of parathyroid
hormone. This hormone acts on the kidney, stimulating it to produce more
1,25-dihydroxycholecalciferol from its precursor 25-hydroxycholecalciferol.
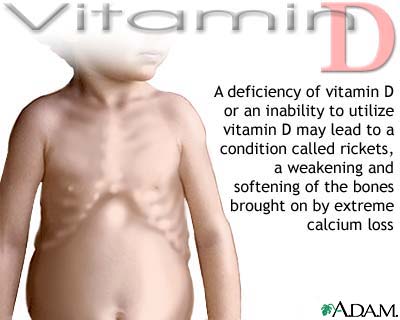
Rickets, a disease of growing bone, is developed
in the deficiency of vitamin D in organism.
http://www.youtube.com/watch?v=n7vybcT9_F4
As with vitamin A, excessive intake of vitamin D causes the bones to become
fragile and to undergo multiple fractures, suggesting that both vitamins play a
role in biological transport and deposition of calcium.

Most natural foods contain little of vitamin D; vitamin D in the diet comes
largely from fish-liver oils, liver, yoke of eggs, butter. Vitamin D
preparations available commercially are products of the ultraviolet irradiation
of ergosterol from yeast.
About 2,5-10 mkg of vitamin D is required by an
adult daily and 12-25 mkg by children. The vitamin
can be stored in sufficient amounts in the liver for a single dose to suffice
for some weeks.
|
|
|
a-Tocopherol |
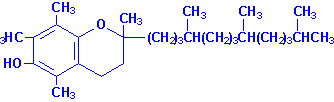
Vitamin E was first recognized
as a factor in vegetable oils that restores fertility in rats grown on cow's
milk alone and otherwise incapable of bearing young. It was isolated from wheat
germ and was given the name tocopherol. Several
different tocopherols having vitamin E activity have
been found in plants; the most active and abundant is a-tocopherol.
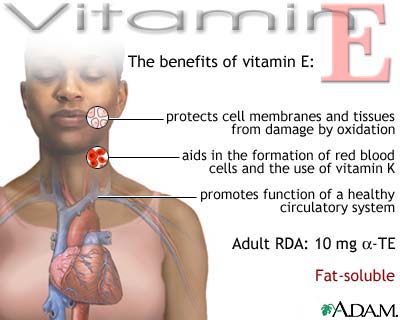
The deficiency of tocopherol produces many other symptoms besides infertility
in male and female, e.g., degeneration of the kidneys, the deposition of brown
pigments in lipid depots, necrosis of the liver, and dystrophy, or wasting, of
skeletal muscles.
Tocopherols have been found to have antioxidant activity; i.e., they prevent the autoxidation of highly unsaturated fatty acids when they
are exposed to molecular oxygen. One of the functions of tocopherol
may be to protect highly unsaturated fatty acids in the lipids of biological
membranes against the deleterious effects of molecular oxygen. Normally, autoxidation products of unsaturated fats do not occur in
the tissues, but in tocopherol deficiency they are
detectable in the fat depots, liver, and other organs.
Due to the hydrophobic side radical tocopherol can be built into the phospholipid
matrix of biological membranes and stabilize the mobility and microviscosity of membrane proteins and lipids.
Tocopherol is the most
potent natural antioxidant.
About 10-20 mg of vitamin E is required per day.
The most abundant sources of vitamin E are oils (sunflower, corn, soybean oils), fresh vegetables, animal stuffs (meat,
butter, egg yoke).
http://www.youtube.com/watch?v=8oRUF_g-J3k&feature=related
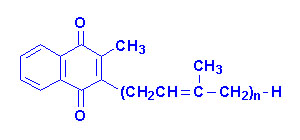
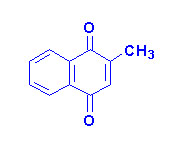
Vitamin K
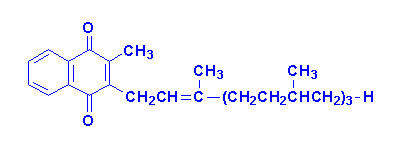
The K vitamins exist
naturally as K1 (phylloquinone) in green vegetables and K2
(menaquinone) produced by intestinal bacteria and K3 is synthetic
menadione. When administered, vitamin K3 is alkylated to one of the
vitamin K2 forms of menaquinone.
|
|
|
|
|
Vitamin K1 |
Vitamin K2 |
Vitamin |
Vitamin K was first discovered as a nutritional factor
required for normal blood-clotting time. At least two forms of vitamin K are
known; vitamin K2 is believed
to be the active form. Vitamin K deficiency cannot readily be produced in rats
and other mammals because the vitamin is synthesized by intestinal bacteria.
http://www.youtube.com/watch?v=DVGsnlVCoeA
http://www.youtube.com/watch?v=WI24c2LYFug&feature=related
The only known result of vitamin K deficiency is
a failure in the biosynthesis of the enzyme proconvertin in the liver. This
enzyme catalyzes a step in a complex sequence of reactions involved in the
formation of prothrombin,
the precursor of thrombin, a protein
that accelerates the conversion of fibrinogen
into fibrin, the insoluble protein
constituting the fibrous portion of blood clots.
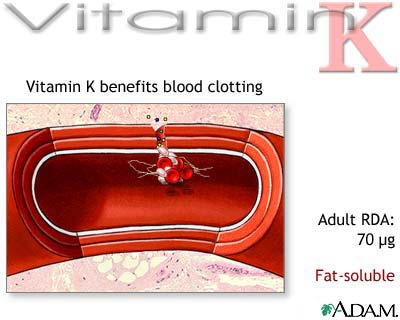
The compound dicumarol, an analog of vitamin K, produces symptoms in animals resembling
vitamin K deficiency; it is believed to block the action of vitamin K. Dicumarol is used in clinical medicine to prevent clotting in
blood vessels. Dicumarol is the antivitamin
of vitamin K.
Some evidence indicates that vitamin K may
function as a coenzyme in a specialized route of electron transport in animal
tissues; since vitamin K is a quinone which can be
reduced reversibly to a quinol, it may serve as an
electron carrier.
Function
- aids in reducing excessive menstrual flow
- aids the absorption of calcium in bone
- essential for normal liver functioning
- essential for synthesis of four proteins that act
in coagulation
- important in maintaining vitality and longevity
- necessary for formation of prothrombin which is
required for effective blood clotting
- involved in electron transport mechanism and
oxdative phosphorylation
Food Source
- alfalfa
- blackstrap
molasses
- broccoli
- Brussels
sprouts
- cauliflower
- cereals
- cow's milk
- egg yolks
- fish liver
oils
- green
plants, such as lettuce
- kelp
- leafy
green vegetables, such as cabbage, spinach
- meats,
such as pig and beef liver
- peas
- polyunsaturated
oils
- potatoes
- string
beans
- yogurt
Effective With
Increased Intakes Needed
- after prolonged paraffin ingestion
- for those with biliary obstructions
- for those with liver disease
- if taking antibiotics for long duration
- if you have a malabsorption disease
- in newborn babies
- in overdose of anticoagulant drugs, such as
Warfarin, Dicoumarol, which neutralize the effect of Vitamin K
Used For
- anticoagulant drug overdose
- hemorrhagic disease in newborn babies
- inhibiting some cancer tumors
- overcoming inability to absorb vitamins
- overcoming effects of antibiotics on intestinal
bacteria
- protection against osteoporosis
Destroyed By
- acids
- alkalis
- commercial
processing
- light and
ultra-violet irradiation
- oxidizing
agents
Symptoms of Deficiency
- excessive bleeding and hemorrhage
In babies:
- bleeding from the stomach, intestines, umbilical
cord site
Deficiency Caused By
In Babies:
- low levels in human breast milk
- poor transfer across placenta
- sterile intestine with no bacteria
In Adults:
- as a consequence of sprue
- Celiac's Disease
- destruction of intestinal bacteria by antibiotics
- lack of bile salts
- liver conditions, such as viral hepatitis
- surgical removal of intestines
- prolonged ingestion of liquid paraffin
Deficiency Leads To
- inability of blood to coagulate
Hypovitaminos of vitamin K in man can be developed in liver diseases when there is the
decrease of bile acids amount in intestine and as result the inhibition of fat
soluble substances absorption is observed.
Vitamin K is produced by many microorganisms in the intestine. also Plants
(cabbage, tomato, lettuce)are natural sources of vitamin K.
Adult person requires 200-300 mkg of vitamin K
per day.
References:
1.
John Mc Murry,
Mary E. Castellion. General, Organic and Biological
Chemistry.- New Jersy: Prentice Hall, 1992.- 764 p.
2.
John W. Suttie.
Introduction to Biochemistry. –
3.
Robert K. Murray, Daryl K. Granner. Harper’s illustrated Biochemistry. –
4.
VK Malhotra.
Biochemistry for students. –
5.
Lehninger A. Principles of Biochemistry. –
6.
Stryer L. Biochemistry. –
Investigation of water soluble
(coenzyme)
vitamins functional role in metabolism and cell functions realization.
Vitamins are nutrients required in tiny amounts for
essential metabolic reactions in the body. The term vitamin
does not include other essential nutrients
such as dietary minerals,
essential fatty acids,
or essential amino
acids, nor does it encompass the large number of other nutrients
that promote health but that are not essential for life.
Vitamins are bio-molecules that act both as catalysts and substrates
in chemical reactions.
When acting as a catalyst, vitamins are bound to enzymes
and are called cofactors.
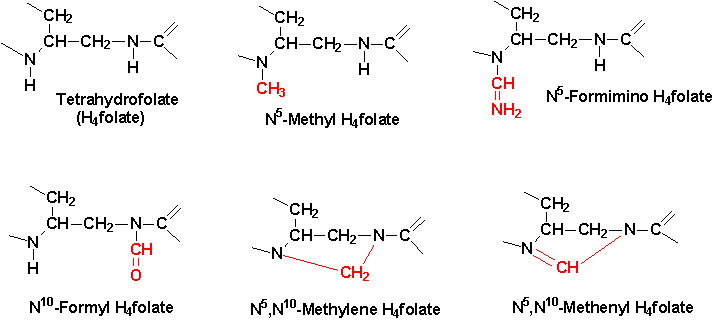
(For example, vitamin K forms part of the proteases involved in blood clotting.) Vitamins also act as coenzymes to carry chemical groups between
enzymes. (For example, folic acid
carries various forms of carbon groups–methyl, formyl
or methylene–in the cell.)/
 Until
the 1900s, vitamins were obtained solely through food intake. Many food sources
contain different ratios of vitamins. Therefore, if the only source of vitamins
is food, changes in diet will alter the types and amounts of vitamins ingested.
However, as many vitamins can be stored by the body, short-term deficiencies
(which, for example, could occur during a particular growing season) do not
usually cause disease.
Until
the 1900s, vitamins were obtained solely through food intake. Many food sources
contain different ratios of vitamins. Therefore, if the only source of vitamins
is food, changes in diet will alter the types and amounts of vitamins ingested.
However, as many vitamins can be stored by the body, short-term deficiencies
(which, for example, could occur during a particular growing season) do not
usually cause disease.
Vitamins have been produced as
commodity chemicals and made widely available as inexpensive pills for several
decades,[2]
allowing supplementation of the dietary intake.
Difference
from water soluble vitamins: water
soluble vitamins are included into coenzymes, don't have provitamins,
are not included into the membranes, and hypervitaminoses are not peculiar for them.
With exception of vitamin B6
and B12, they are readily excreted in urine without appreciable
storage, so frequent consumption becomes necessary. They are generally nontoxic
when present in excess of needs, although symptoms may be reported in people
taking megadoses of niacin, vitamin C, or pyridoxine (vitamin B6). All the B
vitamins function as coenzymes or cofactors, assisting in the activity of
important enzymes and allowing energy-producing reactions to proceed normally. As a result, any lack of water-soluble vitamins mostly affects growing or
rapidly metabolizing tissues such as skin, blood, the digestive tract, and the nervous system. Water-soluble vitamins
are easily lost with overcooking.
|
Water-soluble vitamins and their
characteristics. |
|
|
||||||
|
Common food sources |
Major functions |
Deficiency symptoms |
Overconsumption symptoms |
Stability in foods |
|
|
||
|
Vitamin C (abscorbic acid) |
|
|
||||||
|
Citrus fruits, broccoli, strawberries, melon,
green pepper, tomatoes, dark green vegetables, potatoes. |
Formation of collagen (a component of
tissues), helps hold them together; wound healing; maintaining blood vessels,
bones, teeth; absorption of iron, calcium, folacin;
production of brain hormones, immune factors; antioxidant. |
Bleeding gums; wounds don't heal; bruise
easily; dry, rough skin; scurvy; sore joints and bones; increased infections. |
Nontoxic under normal conditions; rebound
scurvy when high doses discontinued; diarrhea, bloating, cramps; increased
incidence of kidney stones. |
Most unstable under heat, drying, storage;
very soluble in water, leaches out of some vegetables during cooking;
alkalinity (baking soda) destroys vitamin C. |
|
|
||
|
Thiamin (vitamin B1 ) |
|
|
||||||
|
Pork, liver, whole grains, enriched grain
products, peas, meat, legumes. |
Helps release energy from foods; promotes
normal appetite; important in function of nervous system. |
Mental confusion; muscle weakness, wasting;
edema; impaired growth; beriberi. |
None known. |
Losses depend on cooking method, length,
alkalinity of cooking medium; destroyed by sulfite used to treat dried fruits
such as apricots; dissolves in cooking water. |
|
|
||
|
Riboflavin (vitamin B2) |
|
|
||||||
|
Liver, milk, dark green vegetables, whole and
enriched grain products, eggs. |
Helps release energy from foods; promotes good
vision, healthy skin. |
Cracks at corners of mouth; dermatitis around
nose and lips; eyes sensitive to light. |
None known. |
Sensitive to light; unstable in alkaline
solutions. |
|
|||
|
Niacin (nicotinamide, nicotinic
acid) |
|
|
||||||
|
Liver, fish, poultry, meat, peanuts, whole and
enriched grain products. |
Energy production from foods; aids digestion,
promotes normal appetite; promotes healthy skin, nerves. |
Skin disorders; diarrhea; weakness; mental
confusion; irritability. |
Abnormal liver function; cramps; nausea;
irritability. |
|
|
|||
|
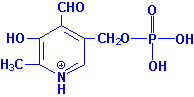
Vitamin B6 (pyridoxine, pyridoxal,
pyridoxamine) |
|
|
||||||
|
Pork, meats, whole grains and cereals,
legumes, green, leafy vegetables. |
Aids in protein metabolism, absorption; aids
in red blood cell formation; helps body use fats. |
Skin disorders, dermatitis, cracks at corners
of mouth; irritability; anemia; kidney stones; nausea; smooth tongue. |
None known. |
Considerable losses during cooking. |
|
|||
|
|
|
|
|
|
|
|
|
|
|
Folacin (folic acid) |
|
||||
|
Liver, kidney, dark green leafy vegetables, meats,
fish, whole grains, fortified grains and cereals, legumes, citrus fruits. |
Aids in protein metabolism; promotes red blood
cell formation; prevents birth defects of spine, brain; lowers homocystein levels and thus coronary heart disease risk. |
Anemia; smooth tongue; diarrhea. |
May mask vitamin B12 deficiency
(pernicious anemia). |
Easily destroyed by storing, cooking and other
processing. |
|
|
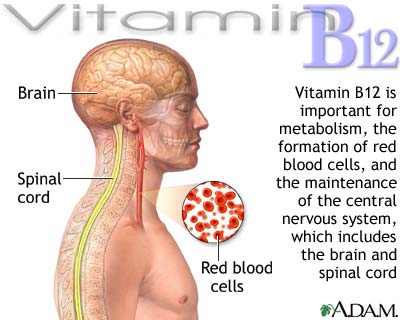
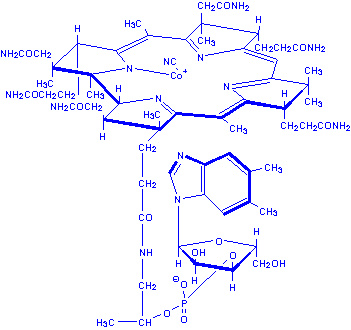
Vitamin B12 |
|
||||
|
Found only in animal foods: meats, liver, kidney,
fish, eggs, milk and milk products, oysters, shellfish. |
Aids in building of genetic material; aids in
development of normal red blood cells; maintenance of nervous system. |
Pernicious anemia, anemia; neurological disorders;
degeneration of peripheral nerves that may cause numbness, tingling in
fingers and toes. |
None known. |
|
|
|
Pantothenic acid |
|
||||
|
Liver, kidney, meats, egg yolk, whole grains,
legumes; also made by intestinal bacteria. |
Involved in energy production; aids in
formation of hormones. |
Uncommon due to availability in most foods;
fatigue; nausea, abdominal cramps; difficulty sleeping. |
None known. |
About half of pantothenic
acid is lost in the milling of grains and heavily refined foods. |
|
Thiamin
(Vitamin B1)
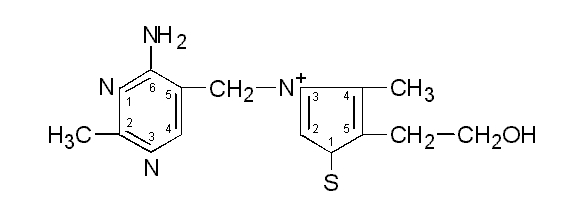
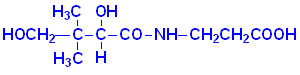
Thiamine or thiamin,
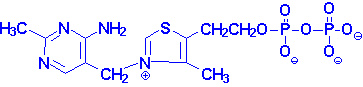
also known as vitamin B1, is a colorless compound with chemical formula C12H17N4OS. It is soluble in water and insoluble in alcohol.
Thiamine decomposes if heated. Its chemical structure
contains a pyrimidine ring and a thiazole ring.Thiamine was first discovered in 1910 by Umetaro Suzuki in Japan when researching how rice bran cured patients of Beriberi. He named it aberic acid. Thiamine
diphosphate (ThDP) or thiamine
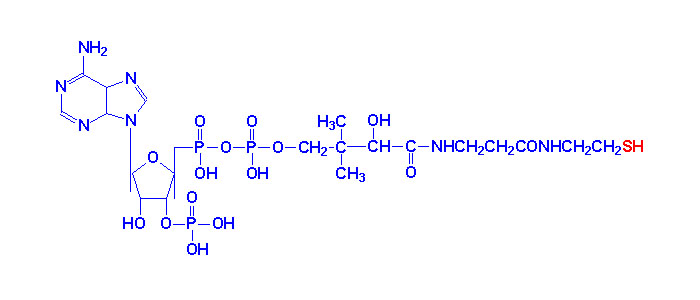
pyrophosphate (TPP) is a coenzyme for pyruvate dehydrogenase, α-ketoglutarate dehydrogenase, branched-chain
Thiamin pyrophosphate
alpha-keto
acid dehydrogenase, and transketolase. The first two of these enzymes function in the metabolism of carbohydrates, while transketolase
functions in the pentose phosphate pathway to synthesize NADPH and the pentose sugars deoxyribose and ribose. In general, TPP functions as
a cofactor for enzymes that catalyze the dehydrogenation (decarboxylation
and subsequent conjugation to Coenzyme A) of alpha-keto
acids. TPP is synthesized by the enzyme thiamine
pyrophosphokinase, which requires free thiamine, magnesium, and adenosine triphosphate.
Good sources: Thiamine is found naturally in the following foods, each of which contains
at least 0.1mg of the vitamin per 28-100g (1-3.5oz): Green peas, Spinach, Liver, Beef, Pork, Navy beans, Nuts, Pinto beans, Soybeans, Whole-grain and Enriched
Cereals, Breads, Yeast, and Legumes. 

http://www.youtube.com/watch?v=Q5BCmsixuqM
Thiamin functions as the coenzyme thiamin pyrophosphate (TPP) in the metabolism of carbohydrate and in conduction of nerve impulses. Thiamin
deficiency causes beri-beri, which is frequently seen
in parts of the world where polished (white) rice or unenriched
white flour are predominantly eaten.
http://www.youtube.com/watch?v=PD_CoEngu4M&feature=related
There are three basic expressions of beriberi:
childhood, wet, and dry. Childhood beriberi stunts growth in infants and
children. Wet beriberi is the classic form, with swelling due to fluid
retention (edema) in the lower limbs
that spreads to the upper body, affecting the heart and leading to heart
failure. Dry beriberi affects peripheral nerves, initially causing tingling or
burning sensations in the lower limbs and progressing to nerve degeneration,
muscle wasting, and weight loss.
Thiamine-deficiency disease in North America commonly occurs in people with
heavy alcohol consumption and is called Wernicke-Korsakoff
syndrome. It is caused by poor food intake and by decreased absorption and increased excretion
caused by alcohol consumption.
Riboflavin (Vitamin B2)
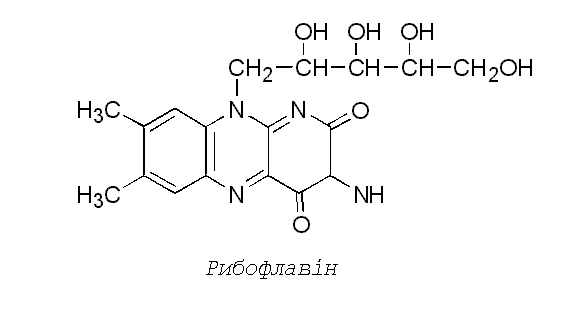
Riboflavin is stable when heated in ordinary cooking,
unless the food is exposed to ultraviolet radiation (sunlight). To prevent
riboflavin breakdown, riboflavin-rich foods such as milk, milk products, and
cereals are packaged in opaque containers. Riboflavin is a component of two
coenzymes—flavin mononucleotide (FMN) and flavin adenine dinucleotide
(FAD)—that act as hydrogen carriers when carbohydrates and fats are used to
produce energy. It is helpful in maintaining good vision and healthy hair, skin
and nails, and it is necessary for normal cell growth.
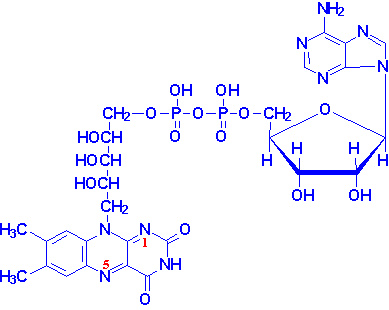
Thiamin pyrophosphate
Riboflavin deficiency causes a
condition known as ariboflavinosis, which is marked
by cheilosis (cracks at the corners of the mouth), oily
scaling of the skin, and a red, sore tongue. In addition, cataracts may occur more frequently with riboflavin deficiency. A
deficiency of this nutrient is
usually a part of multinutrient deficiency and does
not occur in isolation. In North America, it is mostly observed in alcoholics,
elderly persons with low income or depression,
and people with poor eating habits, particularly those who consume
highly refined and fast foods and those who do not consume milk and milk
products.

http://www.youtube.com/watch?v=qpvNaGIJMzw
Unlike fat-soluble vitamins, water-soluble vitamins
are easily lost during cooking and processing. The body does not store excess
quantities of most water-soluble vitamins, so foods bearing them must be
consumed frequently.
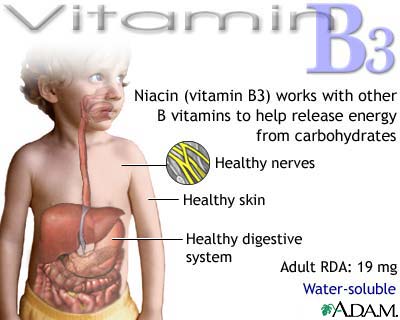
Niacin (Vitamin B5)
|
|
|
|
Nicotinamide |
Nicotinic Acid |
Niacin exists in two forms,
nicotinic acid and nicotinamide. Both forms are
readily absorbed from the stomach and the small intestine. Niacin is stored in
small amounts in the liver and transported to tissues, where it is converted to
coenzyme forms. Any excess is excreted in urine. Niacin is one of the most stable
of the B vitamins. It is resistant to heat and light, and to both acid and
alkali environments. The human body is capable of converting the amino acid tryptophan
to niacin when needed. However, when both tryptophan
and niacin are deficient, tryptophan is used for protein synthesis.
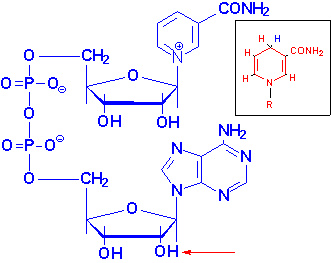
Structure
of NAD+
There are two coenzyme forms
of niacin: nicotinamide adenine dinucleotide
(NAD+) and nicotinamide adenine dinucleotide phophate (NADP+).
They both help break down and utilize proteins, fats, and carbohydrates for
energy. Niacin is essential for growth and is involved in hormone synthesis.
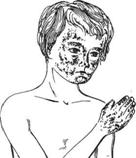
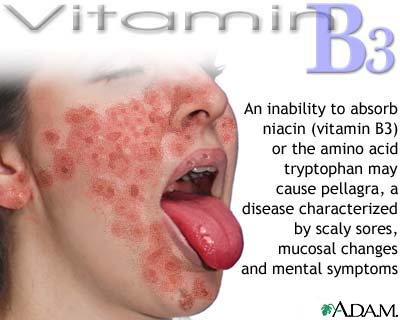
Pellagra results from a
combined deficiency of niacin and tryptophan.
Long-term deficiency leads to central nervous system dysfunction manifested as
confusion, apathy, disorientation, and eventually coma and death. Pellagra is
rarely seen in industrialized countries, where it may be observed in people
with rare disorder of tryptophan metabolism (Hartnup's disease), alcoholics, and those with diseases
that affect food intake.
http://www.youtube.com/watch?v=UrDeVyiXzyg&feature=related
The liver can synthesize niacin from the essential aminoacid
http://www.youtube.com/watch?v=MLFZ8CsrJqU&feature=related
whole grain cereals, legumes, eggs, meat, and royal jelly
http://www.youtube.com/watch?v=o9lJoKoF4DE
- co-enzyme in wide variety of
body metabolic reactions
- needed for production of energy
from carbohydrates, fats and proteins
- needed for interconversions
- essential for maintenance of
healthy skin, hair, sweat glands, nerves, bone marrow and glands producing
sex hormones
- Brewer's Yeast
- cheese
- eggs
- maize
- fish, fatty, white
- meats, especially pig liver and
kidney
- milk
- oats
- wheat bran
- wheat germ
- wholemeal grains
- unpolished brown rice
- vegetables
- yoghurt
- seborrheic dermatitis
- Leiner's Disease
- alopecia (hair falling out in
handfuls)
- scalp disease
- skin complaints
- preventing cot death (given to
babies)
- depression
- diminished reflexes
- fatigue
- hair loss
- increase in blood cholesterol
levels
- loss of appetite
- muscular pains
- nausea
- pale, smooth tongue
- sleepiness
- specific anemia
- deficiency may be induced by
excessive intake of raw egg whites, which contain the protein Avidin which
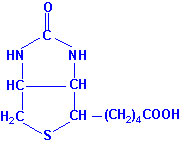
immobilizes Biotin
High quality Vitamin B (Biotin) can be purchased
from Global Herbal Supplies
http://www.youtube.com/watch?v=zwUc5gHoF_U&feature=related
http://www.youtube.com/watch?v=Q9yBs6wvMFc
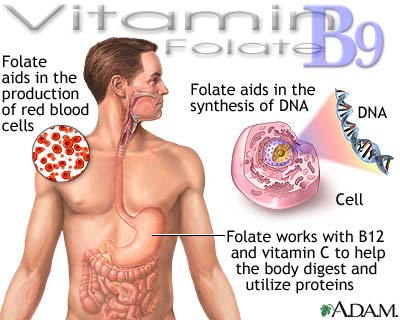
Folic Acid, Folate,
Folacin (Vitamin B9)
- involved in the formation of new
cells
- involved in the metabolism of
ribonucleic acids (RNA) and deoxyribonucleic acids (DNA), essential for
protein synthesis, formation of blood and transmission of genetic code
- essential during pregnancy to
reduce the risk of neural tube defects (birth defects affecting the brain
and/or spinal cord)essential for the normal growth and development of the
fetus
- involved in the biosynthesis of
purines, serines and glycine
- involved in some functions
associated with Vitamin B12
- necessary for building
resistance to diseases in the thymus gland of new born babies and infants
- may reduce the risk of cervical
dysplasia
- necessary for red blood cell
production
- bananas
- Brewers's Yeast
- citrus fruits, peeled
- eggs
- fatty fish
- fresh nuts
- green leafy vegetables
- meats, especially pig liver and
kidney
- milk
- oats
- pulses, such as lentils
- roasted nuts
- soy products, such as tofu
- unpolished brown rice
- wheat germ
- wheat bran
- wheat grains
- by alcohol drinkers
- by the elderly
- during pregnancy and
breastfeeding
- if taking contraceptive pill
- if taking the drugs, Aspirin,
Cholestyramine, Isethionate, Isoniazid, Methotrexate,
Pentamidime, Phenytoin (may be neutralized), Primidone, Pyrimethamine,
Triamterene,Trimethoprim
- malabsorption in geriatric
patients
- megaloblastic anemia
- mental deterioration
- psychosis
- schizophrenia
- leached into cooking water
- processing and cooking of
vegetables, fruits and dairy products
- unstable to oxygen at high
temperatures but protected by Vitamin C
Various conditions relating to
childbirth:
- abortion
- birth defects, such as neural
tube defect which causes spina bifida
- hemorrhage following birth
- premature birth
- premature separation of the
placenta from the uterus
- toxemia
- megaloblastic anemia (red blood
cells are large and uneven with a shortened life span)
- mild mental symptoms, such as
forgetfulness and confusion
Folic Acid has a low toxicity but
occasionally the following symptoms occur:
- abdominal distension
- flatulence (gas/wind)
- irritability
- loss of appetite
- nausea
- over-activity
- sleep disturbance
- symptoms of fever
- temperature rise
Long term high doses may cause
Vitamin B12 losses from the body
http://www.youtube.com/watch?v=4-pMZRxyasU&feature=related
http://www.youtube.com/watch?v=TLDodF9kkRo&feature=related
http://www.youtube.com/watch?v=_QFl7BnWhpQ&feature=related
http://www.youtube.com/watch?v=IQ0Aet8FVVU&feature=related
http://www.youtube.com/watch?v=DQ7IHIgw1ic&feature=related
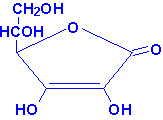
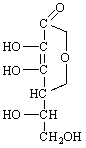
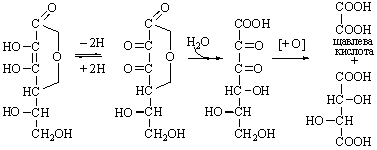
Ascorbic Dehydroascorbic Diketogulonic
Biological role of ascorbic acid:
-
inhibits the oxidation of hemoglobin;
-
accelerates the oxidation of glucose in pentose phosphate
pathway;
-
reduces the disulfide bonds to sulfhydryl
bonds;
-
is necessary for hydroxylation of cholesterol;
-
takes part in metabolism of adrenaline;
-
is necessary for the metabolism of mineral elements (Fe,
Ca);
- accelerates the synthesis of glycogen in liver.
While at sea in May 1747, Lind provided some crewmembers with two oranges
http://www.youtube.com/watch?v=o3uSmyYSBJ8&feature=related
http://www.youtube.com/watch?v=ACBQQOUcn-Y&feature=related
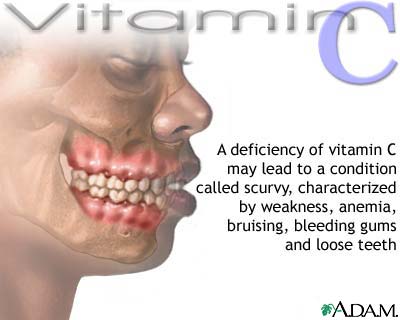
The deficiency of
vitamin P in organism results in the petechias (dot
hemorrhages on skin).