Introduction to
metabolism. Investigation of aerobic and anaerobic oxidation of glucose.
Other ways of monosaccharides metabolism.
Carbohydrates
Carbohydrates have the general molecular formula
CH2O, and thus were once thought to represent "hydrated
carbon". However, the arrangement of atoms in carbohydrates has little to
do with water molecules.
http://www.youtube.com/watch?v=p-lFJVOkFwk
Starch and cellulose are two common
carbohydrates. Both are macromolecules
with molecular weights in the hundreds of thousands. Both are polymers
(hence "polysaccharides"); that is, each is built from
repeating units, monomers,
much as a chain is built from its links.
The monomers of both starch and cellulose are the
same: units of the sugar glucose.
Sugars
Monosaccharides
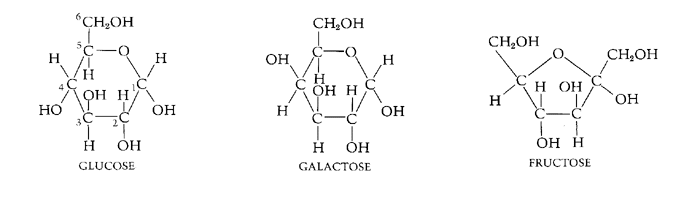 Three common
sugars share the same molecular formula: C6H12O6.
Because of their six carbon atoms, each is a hexose.
Three common
sugars share the same molecular formula: C6H12O6.
Because of their six carbon atoms, each is a hexose.
They
are:
·
glucose, "blood
sugar", the immediate source of energy for cellular respiration
·
galactose, a sugar in
milk (and yogurt), and
·
fructose, a sugar
found in honey.
Although all three share the same molecular
formula (C6H12O6), the arrangement of atoms
differs in each case. Substances such as these three, which have identical
molecular formulas but different structural formulas, are known as structural
isomers.
Glucose, galactose, and fructose are
"single" sugars or monosaccharides. Two monosaccharides can be
linked together to form a "double" sugar or disaccharide.
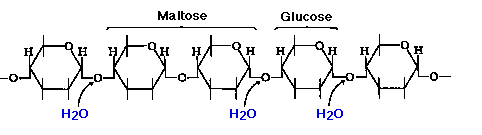
Disaccharides
Three
common disaccharides:
·
sucrose — common
table sugar = glucose + fructose
·
lactose — major
sugar in milk = glucose + galactose
·
maltose — product of
starch digestion = glucose + glucose
Although the process of linking the
two monomers is rather complex, the end result in each case is the loss of a
hydrogen atom (H) from one of the monosaccharides and a hydroxyl group (OH)
from the other. The resulting linkage between the sugars is called a glycosidic
bond. The molecular formula of each of these disaccharides is
C12H22O11 = 2 C6H12O6
− H2O
All sugars are very soluble in water because of their
many hydroxyl groups. Although not as concentrated a fuel as fats, sugars are
the most important source of energy for many cells.
Carbohydrates provide the bulk of the
calories (4 kcal/gram)
in most diets, and starches provide the bulk of that. Starches are
polysaccharides.
Polysaccharides
Starches
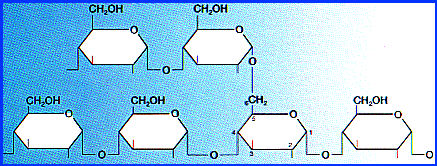
Starches are polymers of glucose. Two
types are found:
·
amylose consists of linear,
unbranched chains of several hundred glucose residues (units). The glucose
residues are linked by a glycosidic bond between their #1 and #4 carbon atoms.
·
amylopectin
differs from amylose in being highly branched. At approximately every thirtieth
residue along the chain, a short side chain is attached by a glycosidic bond to
the #6 carbon atom (the carbon above the ring). The total number of glucose
residues in a molecule of amylopectin is several thousand.
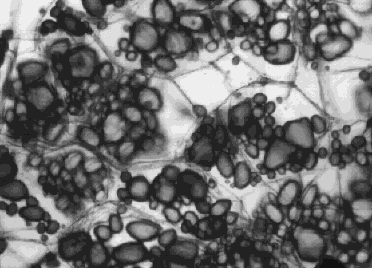
Starches are insoluble in water and thus can
serve as storage depots of glucose. Plants convert excess glucose into starch
for storage. The image shows starch grains (lightly stained with iodine) in the
cells of the white potato. Rice, wheat, and corn are also major sources of
starch in the human diet.
Before starches can enter
(or leave) cells, they must be digested. The hydrolysis of starch is done by
amylases. With the aid of an amylase (such as pancreatic amylase),
water molecules enter at the 1 -> 4 linkages, breaking the chain and
eventually producing a mixture of glucose and maltose. A
different amylase is needed to break the 1 -> 6 bonds of amylopectin.
http://www.youtube.com/watch?v=AEsQxzeAry8
http://www.youtube.com/watch?v=KED6BHVM97s&feature=related
Glycogen
Animals store excess glucose by polymerizing it
to form glycogen. The structure of glycogen is similar to that of
amylopectin, although the branches in glycogen tend to be shorter and more
frequent.
Glycogen is broken back down into glucose when
energy is needed (a process called glycogenolysis).
http://www.youtube.com/watch?v=D2RFc1D_Iv0&feature=related
In glycogenolysis,
·
phosphate groups — not water — break
the 1 -> 4 linkages
·
the phosphate group must then be
removed so that glucose can leave the cell.
The liver and skeletal muscle are major depots of
glycogen.
There is some evidence that intense exercise and
a high-carbohydrate diet ("carbo-loading") can increase the reserves
of glycogen in the muscles and thus may help marathoners work their muscles
somewhat longer and harder than otherwise. But for most of us, carbo loading
leads to increased deposits of fat.
Cellulose
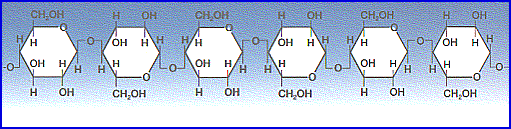
Cellulose is probably the single most abundant
organic molecule in the biosphere. It is the major structural material of which
plants are made. Wood is largely cellulose while cotton and paper are almost
pure cellulose.
Like starch, cellulose is a
polysaccharide with glucose as its monomer. However, cellulose differs profoundly from starch in its
properties.

·
Because of the orientation of the
glycosidic bonds linking the glucose residues, the rings of glucose are
arranged in a flip-flop manner. This produces a long, straight, rigid molecule.
·
There are no side chains in cellulose
as there are in starch. The absence of side chains allows these linear
molecules to lie close together.
·
Because of the many -OH groups, as well
as the oxygen atom in the ring, there are many opportunities for hydrogen bonds
to form between adjacent chains.
The result is a series of stiff, elongated fibrils
— the perfect material for building the cell walls of plants.
This electron micrograph (courtesy of R. D.
Preston) shows the cellulose fibrils in the cell wall of a green alga.
These long, rigid fibrils are a clear reflection of the nature of the cellulose
molecules of which they are composed.
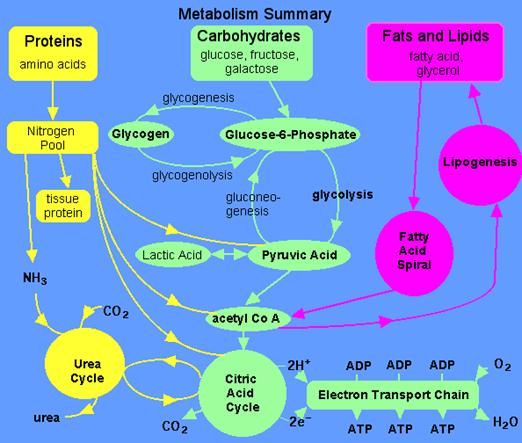
Carbohydrates and Metabolism Chart - Carb Chart
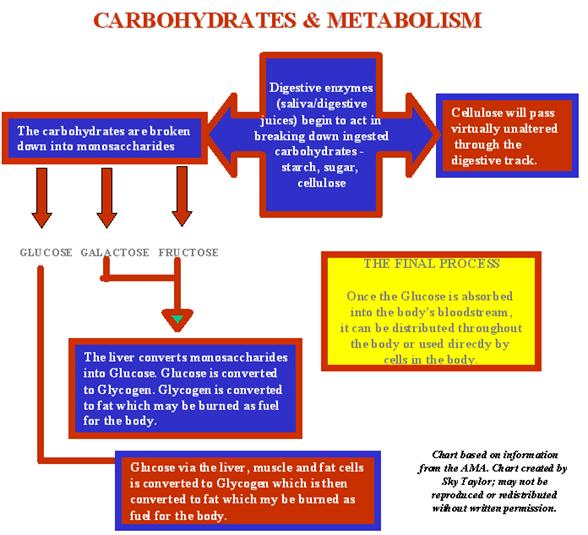



Digestion of
Dietary Carbohydrates
Dietary carbohydrate from which humans gain energy
enter the body in complex forms, such as disaccharides and the polymers starch (amylose and amylopectin) and glycogen. The polymer cellulose is also consumed but not
digested. The first step in the metabolism of digestible carbohydrate is the
conversion of the higher polymers to simpler, soluble forms that can be
transported across the intestinal wall and delivered to the tissues. The
breakdown of polymeric sugars begins in the mouth. Saliva has a slightly acidic
pH of 6.8 and contains lingual amylase that begins the digestion of
carbohydrates. The action of lingual amylase is limited to the area of the
mouth and the esophagus; it is virtually inactivated by the much stronger acid
pH of the stomach. Once the food has arrived in the stomach, acid hydrolysis
contributes to its degradation; specific gastric proteases and lipases aid this
process for proteins and fats, respectively. The mixture of gastric secretions,
saliva, and food, known collectively as chyme, moves to the small
intestine.
The main polymeric-carbohydrate digesting enzyme of
the small intestine is -amylase. This enzyme is secreted by
the pancreas and has the same activity as salivary amylase, producing
disaccharides and trisaccharides. The latter are converted to monosaccharides
by intestinal saccharidases, including maltases that hydrolyze di- and
trisaccharides, and the more specific disaccharidases, sucrase, lactase, and
trehalase. The net result is the almost complete conversion of digestible
carbohydrate to its constituent monosaccharides. The resultant glucose and
other simple carbohydrates are transported across the intestinal wall to the
hepatic portal vein and then to liver parenchymal cells and other tissues.
There they are converted to fatty acids, amino acids, and glycogen, or else
oxidized by the various catabolic pathways of cells.
Oxidation of glucose is known as glycolysis.Glucose is oxidized to
either lactate or pyruvate. Under aerobic conditions, the dominant product in
most tissues is pyruvate and the
pathway is known as aerobic glycolysis.
When oxygen is depleted, as for instance during prolonged vigorous exercise,
the dominant glycolytic product in many tissues is lactate and the process is known as anaerobic glycolysis.
back to the
top
The Energy
Derived from Glucose Oxidation
Aerobic glycolysis of glucose to
pyruvate, requires two equivalents of ATP to activate the process, with the
subsequent production of four equivalents of ATP and two equivalents of NADH.
Thus, conversion of one mole of glucose to two moles of pyruvate is accompanied
by the net production of two moles each of ATP and NADH.
Glucose + 2 ADP + 2 NAD+ + 2 Pi
-----> 2 Pyruvate + 2 ATP + 2 NADH + 2 H+
The NADH generated during glycolysis
is used to fuel mitochondrial ATP synthesis via oxidative
phosphorylation, producing either two or three equivalents of ATP depending
upon whether the glycerol
phosphate shuttle or the malate-aspartate
shuttle is used to transport the electrons from cytoplasmic NADH into the
mitochondria. The net yield from the oxidation of 1 mole of glucose to 2 moles
of pyruvate is, therefore, either 6 or 8 moles of ATP. Complete oxidation of
the 2 moles of pyruvate, through the TCA cycle,
yeilds an additional 30 moles of ATP; the total yield, therefore being either
36 or 38 moles of ATP from the complete oxidation of 1 mole of glucose to CO2
and H2O.
back to the
top
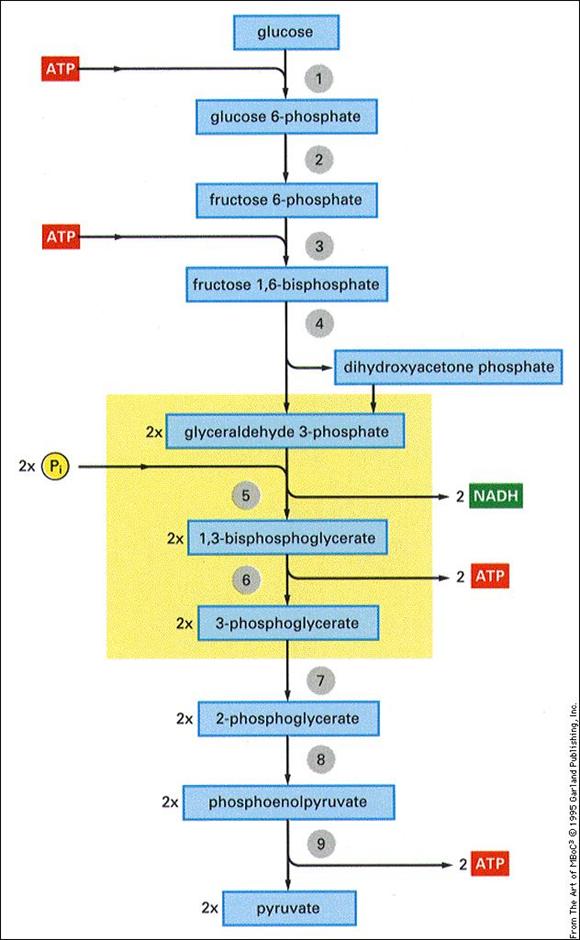
The
Individual Reactions of Glycolysis
The pathway of glycolysis can be seen
as consisting of 2 separate phases. The first is the chemical priming phase
requiring energy in the form of ATP, and the second is considered the
energy-yielding phase. In the first phase, 2 equivalents of ATP are used to
convert glucose to fructose 1,6-bisphosphate (F1,6BP). In the second phase
F1,6BP is degraded to pyruvate, with the production of 4 equivalents of ATP and
2 equivalents of NADH.
http://www.youtube.com/watch?v=6JGXayUyNVw&feature=related
http://www.youtube.com/watch?v=nKgUBsC4Oyo&feature=related
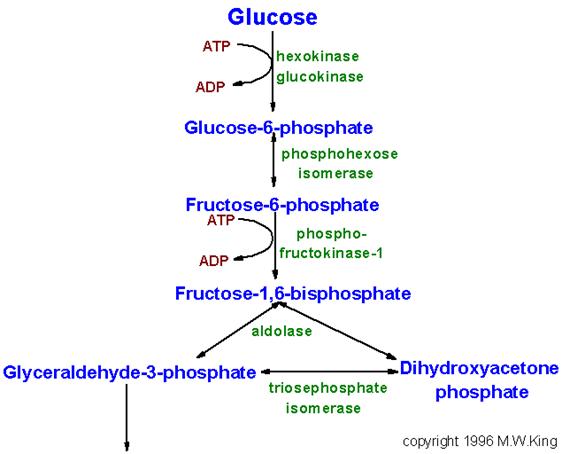
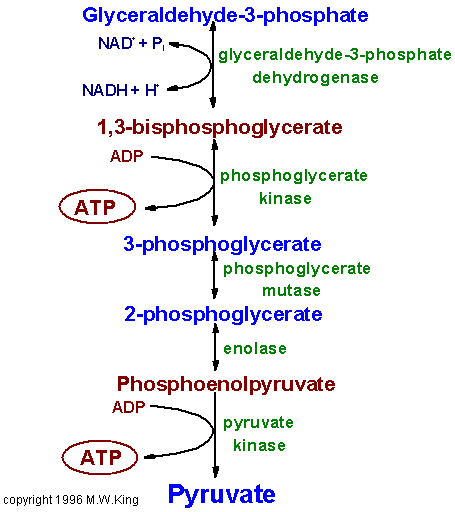
Pathway of glycolysis from glucose to
pyruvate. Substrates and products are in blue, enzymes are in green. The two
high energy intermediates whose oxidations are coupled to ATP synthesis are shown
in red (1,3-bisphosphoglycerate and phosphoenolpyruvate).
http://www.youtube.com/watch?v=PQMsJSme780&feature=related
The Hexokinase Reaction:
The ATP-dependent phosphorylation of
glucose to form glucose 6-phosphate (G6P)is the first reaction of glycolysis,
and is catalyzed by tissue-specific isoenzymes known as hexokinases. The
phosphorylation accomplishes two goals: First, the hexokinase reaction converts
nonionic glucose into an anion that is trapped in the cell, since cells lack
transport systems for phosphorylated sugars. Second, the otherwise biologically
inert glucose becomes activated into a labile form capable of being further
metabolized.
Four mammalian isozymes of hexokinase
are known (Types I - IV), with the Type IV isozyme often referred to as
glucokinase. Glucokinase is the form of the enzyme found in hepatocytes. The
high Km of glucokinase for glucose means that this enzyme is saturated
only at very high concentrations of substrate.
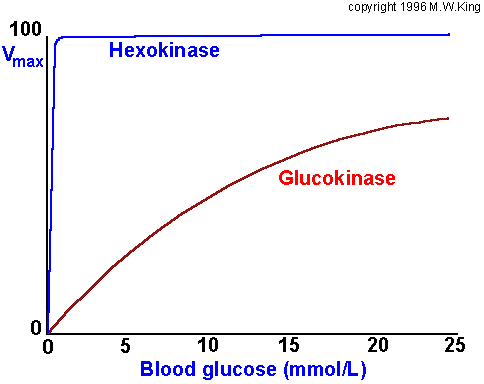
Comparison of the activities of
hexokinase and glucokinase. The Km for hexokinase is significantly
lower (0.1mM) than that of glucokinase (10mM). This difference ensures that
non-hepatic tissues (which contain hexokinase) rapidly and efficiently trap
blood glucose within their cells by converting it to glucose-6-phosphate. One
major function of the liver is to deliver glucose to the blood and this in
ensured by having a glucose phosphorylating enzyme (glucokinase) whose Km
for glucose is sufficiently higher that the normal circulating concentration of
glucose (5mM).
This feature of hepatic glucokinase
allows the liver to buffer blood
glucose. After meals, when postprandial blood glucose levels are high, liver
glucokinase is significantly active, which causes the liver preferentially to
trap and to store circulating glucose. When blood glucose falls to very low
levels, tissues such as liver and kidney, which contain glucokinases but are
not highly dependent on glucose, do not continue to use the meager glucose
supplies that remain available. At the same time, tissues such as the brain,
which are critically dependent on glucose, continue to scavenge blood glucose
using their low Km hexokinases, and as a consequence their viability
is protected. Under various conditions of glucose deficiency, such as long
periods between meals, the liver is stimulated to supply the blood with glucose
through the pathway of gluconeogenesis.
The levels of glucose produced during gluconeogenesis are insufficient to
activate glucokinase, allowing the glucose to pass out of hepatocytes and into
the blood.
The regulation of hexokinase and
glucokinase activities is also different. Hexokinases I, II, and III are
allosterically inhibited by product (G6P) accumulation, whereas glucokinases
are not. The latter further insures liver accumulation of glucose stores during
times of glucose excess, while favoring peripheral glucose utilization when
glucose is required to supply energy to peripheral tissues.
Phosphohexose Isomerase:
The second reaction of glycolysis is an isomerization,
in which G6P is converted to fructose 6-phosphate (F6P). The enzyme catalyzing
this reaction is phosphohexose isomerase (also known as phosphoglucose
isomerase). The reaction is freely reversible at normal cellular concentrations
of the two hexose phosphates and thus catalyzes this interconversion during
glycolytic carbon flow and during gluconeogenesis.
6-Phosphofructo-1-Kinase (Phosphofructokinase-1, PFK-1):
The next reaction of glycolysis involves the
utilization of a second ATP to convert F6P to fructose 1,6-bisphosphate
(F1,6BP). This reaction is catalyzed by 6-phosphofructo-1-kinase, better known
as phosphofructokinase-1 or PFK-1. This reaction is not readily
reversible because of its large positive free energy (G0'
= +5.4 kcal/mol) in the reverse direction. Nevertheless, fructose units readily
flow in the reverse (gluconeogenic) direction because of the ubiquitous
presence of the hydrolytic enzyme, fructose-1,6-bisphosphatase (F-1,6-BPase).
The presence of these two enzymes in the same cell
compartment provides an example of a metabolic futile cycle, which if
unregulated would rapidly deplete cell energy stores. However, the activity of
these two enzymes is so highly regulated that PFK-1 is considered to be the rate-limiting enzyme of glycolysis and
F-1,6-BPase is considered to be the rate-limiting
enzyme in gluconeogenesis.
Aldolase:
Aldolase catalyses the hydrolysis of F1,6BP into two
3-carbon products: dihydroxyacetone phosphate (DHAP) and glyceraldehyde
3-phosphate (G3P). The aldolase reaction proceeds readily in the reverse
direction, being utilized for both glycolysis and gluconeogenesis.
Triose Phosphate Isomerase: \
The two products of the aldolase reaction equilibrate readily in a
reaction catalyzed by triose phosphate isomerase. Succeeding reactions of
glycolysis utilize G3P as a substrate; thus, the aldolase reaction is pulled in
the glycolytic direction by mass action principals.
Glyceraldehyde-3-Phosphate Dehydrogenase:
The second phase of glucose catabolism features the energy-yielding
glycolytic reactions that produce ATP and NADH. In the first of these
reactions, glyceraldehyde-3-P dehydrogenase (G3PDH) catalyzes the NAD+-dependent
oxidation of G3P to 1,3-bisphosphoglycerate (1,3BPG) and NADH. The G3PDH
reaction is reversible, and the same enzyme catalyzes the reverse reaction
during gluconeogenesis.
Phosphoglycerate Kinase:
The high-energy phosphate of 1,3-BPG is used to form
ATP and 3-phosphoglycerate (3PG) by the enzyme phosphoglycerate kinase. Note
that this is the only reaction of glycolysis or gluconeogenesis that involves
ATP and yet is reversible under normal cell conditions. Associated with the
phosphoglycerate kinase pathway is an important reaction of erythrocytes, the
formation of 2,3-bisphosphoglycerate, 2,3BPG (see Figure below) by the enzyme
bisphosphoglycerate mutase. 2,3BPG is an important regulator of hemoglobin's
affinity for oxygen. Note that 2,3-bisphosphoglycerate phosphatase degrades
2,3BPG to 3-phosphoglycerate, a normal intermediate of glycolysis. The 2,3BPG
shunt thus operates with the expenditure of 1 equivalent of ATP per triose
passed through the shunt. The process is not reversible under physiological conditions.
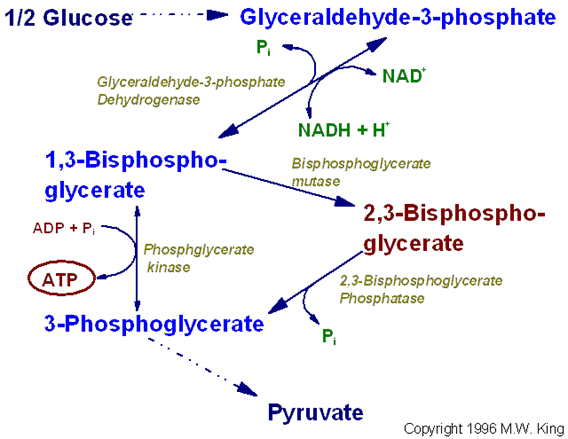
The pathway for 2,3-bisphosphoglycerate (2,3-BPG) synthesis within
erythrocytes. Synthesis of 2,3-BPG represents a major reaction pathway for the
consumption of glucose in erythrocytes. The synthesis of 2,3-BPG in
erythrocytes is critical for controlling hemoglobin affinity for oxygen. Note
that when glucose is oxidized by this pathway the erythrocyte loses the ability
to gain 2 moles of ATP from glycolytic oxidation of 1,3-BPG to
3-phosphoglycerate via the phosphoglycerate kinase reaction.
Phosphoglycerate Mutase and Enolase:
The remaining reactions of glycolysis are aimed at
converting the relatively low energy phosphoacyl-ester of 3PG to a high-energy
form and harvesting the phosphate as ATP. The 3PG is first converted to 2PG by
phosphoglycerate mutase and the 2PG conversion to phosphoenoylpyruvate (PEP) is
catalyzed by enolase
Pyruvate Kinase:
The final reaction of aerobic
glycolysis is catalyzed by the highly regulated enzyme pyruvate kinase (PK). In
this strongly exergonic reaction, the high-energy phosphate of PEP is conserved
as ATP. The loss of phosphate by PEP leads to the production of pyruvate in an
unstable enol form, which spontaneously tautomerizes to the more stable, keto
form of pyruvate. This reaction contributes a large proportion of the free
energy of hydrolysis of PEP.
Anaerobic
Glycolysis
http://www.youtube.com/watch?v=uCmNQQWlrc0&feature=related
Under aerobic conditions, pyruvate in
most cells is further metabolized via the TCA cycle. Under
anaerobic conditions and in erythrocytes under aerobic conditions, pyruvate is
converted to lactate by the enzyme lactate dehydrogenase (LDH), and the lactate
is transported out of the cell into the circulation. The conversion of pyruvate
to lactate, under anaerobic conditions, provides the cell with a mechanism for
the oxidation of NADH (produced during the G3PDH reaction) to NAD+;
which occurs during the LDH catalyzed reaction. This reduction is required
since NAD+ is a necessary substrate for G3PDH, without which
glycolysis will cease. Normally, during aerobic glycolysis the electrons of
cytoplasmic NADH are transferred to mitochondrial carriers of the oxidative
phosphorylation pathway generating a continuous pool of cytoplasmic NAD+.
Aerobic glycolysis generates
substantially more ATP per mole of glucose oxidized than does anaerobic
glycolysis. The utility of anaerobic glycolysis, to a muscle cell when it needs
large amounts of energy, stems from the fact that the rate of ATP production
from glycolysis is approximately 100X faster than from oxidative
phosphorylation. During exertion muscle cells do not need to energize anabolic
reaction pathways. The requirement is to generate the maximum amount of ATP,
for muscle contraction, in the shortest time frame. This is why muscle cells
derive almost all of the ATP consumed during exertion from anaerobic
glycolysis.
The reactions catalyzed by
hexokinase, PFK-1 and PK all proceed with a relatively large free energy
decrease. These nonequilibrium reactions of glycolysis would be ideal
candidates for regulation of the flux through glycolysis. Indeed, in vitro
studies have shown all three enzymes to be allosterically controlled.
Regulation of hexokinase, however, is
not the major control point in glycolysis. This is due to the fact that large amounts
of G6P are derived from the breakdown of glycogen (the predominant mechanism of
carbohydrate entry into glycolysis in skeletal muscle) and, therefore, the
hexokinase reaction is not necessary. Regulation of PK is important for
reversing glycolysis when ATP is high in order to activate gluconeogenesis. As
such this enzyme catalyzed reaction is not a major control point in glycolysis.
The rate limiting step in glycolysis is the reaction catalyzed by PFK-1.
PFK-1 is a tetrameric enzyme that
exist in two conformational states termed R and T that are in equilibrium. ATP
is both a substrate and an allosteric inhibitor of PFK-1. Each subunit has two
ATP binding sites, a substrate site and an inhibitor site. The substrate site
binds ATP equally well when the tetramer is in either conformation. The
inhibitor site binds ATP essentially only when the enzyme is in the T state.
F6P is the other substrate for PFK-1 and it also binds preferentially to the R
state enzyme. At high concentrations of ATP, the inhibitor site becomes
occupied and shifting the equilibrium of PFK-1 comformation to that of the T
state decreasing PFK-1's ability to bind F6P. The inhibition of PFK-1 by ATP is
overcome by AMP which binds to the R state of the enzyme and, therefore,
stabilizes the conformation of the enzyme capable of binding F6P. The most
important allosteric regulator of both glycolysis and gluconeogenesis is fructose 2,6-bisphosphate, F2,6BP,
which is not an intermediate in glycolysis or in gluconeogenesis.
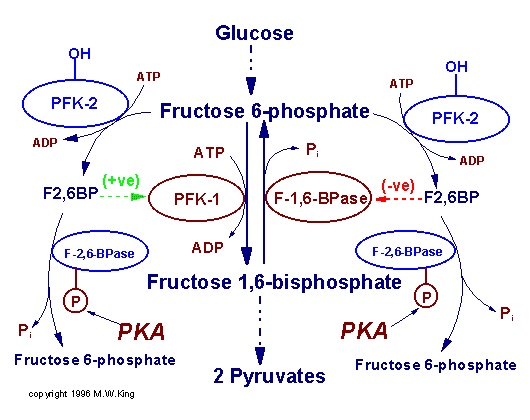
Regulation of
glycolysis and gluconeogenesis by fructose
2,6-bisphosphate (F2,6BP). The major sites for regulation of glycolysis
and gluconeogenesis are the phosphofructokinase-1 (PFK-1) and
fructose-1,6-bisphosphatase (F-1,6-BPase) catalyzed reactions. PFK-2 is the
kinase activity and F-2,6-BPase is the phosphatase activity of the
bi-functional regulatory enzyme,
phosphofructokinase-2/fructose-2,6-bisphosphatase. PKA is cAMP-dependent
protein kinase which phosphorylates PFK-2/F-2,6-BPase turning on the
phosphatase activity. (+ve) and (-ve) refer to positive and negative
activities, respectively.
The synthesis of F2,6BP is catalyzed
by the bifunctional enzyme phosphofructokinase-2/fructose-2,6-bisphosphatase
(PFK-2/F-2,6-BPase). In the nonphosphorylated form the enzyme is known as PFK-2
and serves to catalyze the synthesis of F2,6BP by phosphorylating fructose
6-phosphate. The result is that the activity of PFK-1 is greatly stimulated and
the activity of F-1,6-BPase is greatly inhibited.
Under conditions where PFK-2 is active,
fructose flow through the PFK-1/F-1,6-BPase reactions takes place in the
glycolytic direction, with a net production of F1,6BP. When the bifunctional
enzyme is phosphorylated it no longer exhibits kinase activity, but a new
active site hydrolyzes F2,6BP to F6P and inorganic phosphate. The metabolic
result of the phosphorylation of the bifunctional enzyme is that allosteric
stimulation of PFK-1 ceases, allosteric inhibition of F-1,6-BPase is
eliminated, and net flow of fructose through these two enzymes is
gluconeogenic, producing F6P and eventually glucose.
The interconversion of the
bifunctional enzyme is catalyzed by cAMP-dependent protein kinase (PKA), which
in turn is regulated by circulating peptide hormones. When blood glucose levels
drop, pancreatic insulin production falls, glucagon secretion is stimulated,
and circulating glucagon is highly increased. Hormones such as glucagon bind to
plasma membrane receptors on liver cells, activating membrane-localized
adenylate cyclase leading to an increase in the conversion of ATP to cAMP (see
diagram below). cAMP binds to the regulatory subunits of PKA, leading to
release and activation of the catalytic subunits. PKA phosphorylates numerous
enzymes, including the bifunctional PFK-2/F-2,6-BPase. Under these conditions
the liver stops consuming glucose and becomes metabolically gluconeogenic,
producing glucose to reestablish normoglycemia.
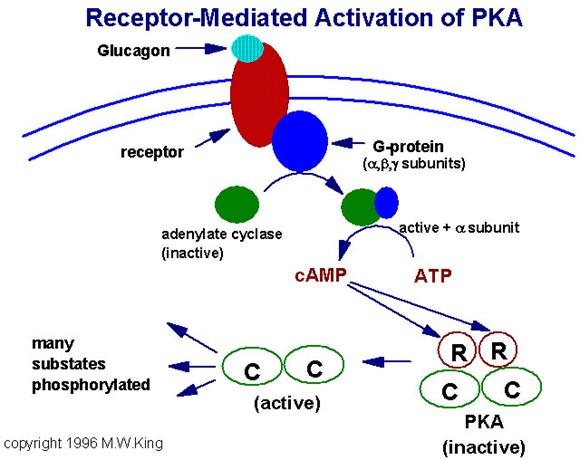
Representative pathway for the
activation of cAMP-dependent protein
kinase (PKA). In this example glucagon binds to its' cell-surface
receptor, thereby activating the receptor. Activation of the receptor is
coupled to the activation of a receptor-coupled G-protein (GTP-binding and
hydrolyzing protein) composed of 3 subunits. Upon activation the alpha subunit
dissociates and binds to and activates adenylate cyclase. Adenylate cylcase
then converts ATP to cyclic-AMP (cAMP). The cAMP thus produced then binds to
the regulatory subunits of PKA leading to dissociation of the associated
catalytic subunits. The catalytic subunits are inactive until dissociated from
the regulatory subunits. Once released the catalytic subunits of PKA
phosphorylate numerous substrate using ATP as the phosphate donor.
Regulation of glycolysis also occurs at the step
catalyzed by pyruvate kinase, (PK). The liver enzyme has been most studied in
vitro. This enzyme is inhibited by ATP and acetyl-CoA and is activated by
F1,6BP. The inhibition of PK by ATP is similar to the effect of ATP on PFK-1.
The binding of ATP to the inhibitor site reduces its affinity for PEP. The
liver enzyme is also controlled at the level of synthesis. Increased
carbohydrate ingestion induces the synthesis of PK resulting in elevated
cellular levels of the enzyme.
A number of PK isozymes have been described. The liver
isozyme (L-type), characteristic of a gluconeogenic tissue, is regulated via
phosphorylation by PKA, whereas the M-type isozyme found in brain, muscle, and
other glucose requiring tissue is unaffected by PKA. As a consequence of these
differences, blood glucose levels and associated hormones can regulate the
balance of liver gluconeogenesis and glycolysis while muscle metabolism remains
unaffected.
In erythrocytes, the fetal PK isozyme has much greater
activity than the adult isozyme; as a result, fetal erythrocytes have
comparatively low concentrations of glycolytic intermediates. Because of the
low steady-state concentration of fetal 1,3BPG, the 2,3BPG shunt (see diagram
above) is greatly reduced in fetal cells and little 2,3BPG is formed. Since
2,3BPG is a negative effector of hemoglobin affinity for oxygen, fetal
erythrocytes have a higher oxygen affinity than maternal erythrocytes.
Therefore, transfer of oxygen from maternal hemoglobin to fetal hemoglobin is
favored, assuring the fetal oxygen supply. In the newborn, an erythrocyte
isozyme of the M-type with comparatively low PK activity displaces the fetal
type, resulting in an accumulation of glycolytic intermediates. The increased
1,3BPG levels activate the 2,3BPG shunt, producing 2,3BPG needed to regulate
oxygen binding to hemoglobin.
Genetic diseases of adult erythrocyte PK are known in
which the kinase is virtually inactive. The erythrocytes of affected
individuals have a greatly reduced capacity to make ATP and thus do not have
sufficient ATP to perform activities such as ion pumping and maintaining
osmotic balance. These erythrocytes have a short half-life, lyse readily, and
are responsible for some cases of hereditary
hemolytic anemia.
The liver PK isozyme is regulated by phosphorylation,
allosteric effectors, and modulation of gene expression. The major allosteric
effectors are F1,6BP, which stimulates PK activity by decreasing its Km(app)
for PEP, and for the negative effector, ATP. Expression of the liver PK gene is
strongly influenced by the quantity of carbohydrate in the diet, with
high-carbohydrate diets inducing up to 10-fold increases in PK concentration as
compared to low carbohydrate diets. Liver PK is phosphorylated and inhibited by
PKA, and thus it is under hormonal control similar to that described earlier
for PFK-2.
Muscle PK (M-type) is not regulated by the same
mechanisms as the liver enzyme. Extracellular conditions that lead to the
phosphorylation and inhibition of liver PK, such as low blood glucose and high
levels of circulating glucagon, do not inhibit the muscle enzyme. The result of
this differential regulation is that hormones such as glucagon and epinephrine
favor liver gluconeogenesis by inhibiting liver glycolysis, while at the same
time, muscle glycolysis can proceed in accord with needs directed by
intracellular conditions.
Pyruvate is the branch point molecule of glycolysis.
The ultimate fate of pyruvate depends on the oxidation state of the cell. In
the reaction catalyzed by G3PDH a molecule of NAD+ is reduced to
NADH. In order to maintain the re-dox state of the cell, this NADH must be
re-oxidized to NAD+. During aerobic glycolysis this occurs in the
mitochondrial electron transport chain generating ATP. Thus, during aerobic
glycolysis ATP is generated from oxidation of glucose directly at the PGK and
PK reactions as well as indirectly by re-oxidation of NADH in the oxidative
phosphorylation pathway. Additional NADH molecules are generated during the
complete aerobic oxidation of pyruvate in the TCA cycle.
Pyruvate enters the TCA cycle in the form of acetyl-CoA which is the product of the pyruvate dehydrogenase
reaction. The fate of pyruvate during anaerobic glycolysis is reduction to
lactate.
During anaerobic glycolysis, that period of time when
glycolysis is proceeding at a high rate (or in anaerobic organisms), the
oxidation of NADH occurs through the reduction of an organic substrate.
Erythrocytes and skeletal muscle (under conditions of exertion) derive all of
their ATP needs through anaerobic glycolysis. The large quantity of NADH
produced is oxidized by reducing pyruvate to lactate. This reaction is carried
out by lactate dehydrogenase, (LDH). The lactate produced during anaerobic
glycolysis diffuses from the tissues and is transproted to highly aerobic
tissues such as cardiac muscle and liver. The lactate is then oxidized to pyruvate
in these cells by LDH and the pyruvate is further oxidized in the TCA cycle. If
the energy level in these cells is high the carbons of pyruvate will be
diverted back to glucose via the gluconeogenesis pathway.
Mammalian cells contain two distinct types of LDH
subunits, termed M and H. Combinations of these different subunits generates
LDH isozymes with different characteristics. The H type subunit predominates in
aerobic tissues such as heart muscle (as the H4 tetramer) while the M subunit
predominates in anaerobic tissues such as skeletal muscle as the M4 tetramer).
H4 LDH has a low Km for pyruvate and also is inhibited by high
levels of pyruvate. The M4 LDH enzyme has a high Km for pyruvate and
is not inhibited by pyruvate. This suggsts that the H-type LDH is utilized for
oxidizing lactate to pyruvate and the M-type the reverse.
Ethanol Metabolism
Animal cells (primarily hepatocytes) contain the
cytosolic enzyme alcohol dehydrogenase (ADH) which oxidizes ethanol to
acetaldehyde. Acetaldehyde then enters the mitochondria where it is oxidized to
acetate by acetaldehyde dehydrogenase (AcDH).
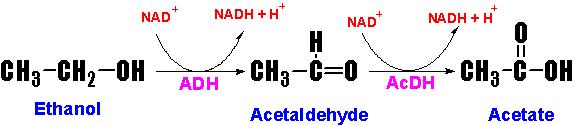
Acetaldehyde forms adducts with proteins, nucleic
acids and other compounds, the results of which are the toxic side effects (the
hangover) that are associated with
alcohol consumption. The ADH and AcDH catalyzed reactions also leads to the
reduction of NAD+ to NADH. The metabolic effects of ethanol
intoxication stem from the actions of ADH and AcDH and the resultant cellular
imbalance in the NADH/NAD+. The NADH produced in the cytosol by ADH
must be reduced back to NAD+ via either the malate-aspartate
shuttle or the glycerol-phosphate
shuttle. Thus, the ability of an individual to metabolize ethanol is
dependent upon the capacity of hepatocytes to carry out eother of these 2
shuttles, which in turn is affected by the rate of the TCA cycle in the
mitochondria whose rate of function is being impacted by the NADH produced by
the AcDH reaction. The reduction in NAD+ impairs the flux of glucose
through glycolysis at the glyceraldehyde-3-phosphate dehydrogenase reaction,
thereby limiting energy production. Additionally, there is an increased rate of
hepatic lactate production due to the effect of increased NADH on direction of
the hepatic lactate dehydrogenase (LDH) reaction. This reverseral of the LDH
reaction in hepatocytes diverts pyruvate from gluconeogenesis leading to a
reduction in the capacity of the liver to deliver glucose to the blood.
In addition to the negative effects of the altered
NADH/NAD+ ratio on hepatic gluconeogenesis, fatty acid oxidation is
also reduced as this process requires NAD+ as a cofactor. In fact
the opposite is true, fatty acid synthesis is increased and there is an
increase in triacylglyceride production by the liver. In the mitocondria, the
production of acetate from acetaldehyde leads to increased levels of
acetyl-CoA. Since the increased generation of NADH also reduces the activity of
the TCA cycle, the acetyl-CoA is diverted to fatty acid synthesis. The
reduction in cytosolic NAD+ leads to reduced activity of
glycerol-3-phosphate dehydrogenase (in the glcerol 3-phosphate to DHAP
direction) resulting in increased levels of glycerol 3-phosphate which is the
backbone for the synthesis of the triacylglycerides. Both of these two events
lead to fatty acid deposition in the liver leading to fatty liver syndrome.
Regulation
of Blood Glucose Levels
If for no other reason, it is because
of the demands of the brain for oxidizable glucose that the human body
exquisitely regulates the level of glucose circulating in the blood. This level
is maintained in the range of 5mM.
Nearly all carbohydrates ingested in
the diet are converted to glucose following transport to the liver. Catabolism
of dietary or cellular proteins generates carbon atoms that can be utilized for
glucose synthesis via gluconeogenesis.
Additionally, other tissues besides the liver that incompletely oxidize glucose
(predominantly skeletal muscle and erythrocytes) provide lactate that can be
converted to glucose via gluconeogenesis.
Maintenance of blood glucose
homeostasis is of paramount importance to the survival of the human organism.
The predominant tissue responding to signals that indicate reduced or elevated
blood glucose levels is the liver. Indeed, one of the most important functions
of the liver is to produce glucose for the circulation. Both elevated and
reduced levels of blood glucose trigger hormonal responses to initiate pathways
designed to restore glucose homeostasis. Low blood glucose triggers release of glucagon from pancreatic -cells.
High blood glucose triggers release of insulin
from pancreatic -cells.
Additional signals, ACTH and growth
hormone, released from the pituitary act to increase blood glucose by inhibiting
uptake by extrahepatic tissues. Glucocorticoids
also act to increase blood glucose levels by inhibiting glucose uptake. Cortisol, the major glucocorticoid
released from the adrenal cortex, is secreted in response to the increase in
circulating ACTH. The adrenal medullary hormone, epinephrine, stimulates production of glucose by activating
glycogenolysis in response to stressful stimuli.
Glucagon binding to its' receptors on
the surface of liver cells triggers an increase in cAMP production leading to
an increased rate of glycogenolysis
by activating glycogen phosphorylase via the PKA-mediated cascade. This is the
same response hepatocytes have to epinephrine release. The resultant increased
levels of G6P in hepatocytes is hydrolyzed to free glucose, by
glucose-6-phosphatase, which then diffuses to the blood. The glucose enters
extrahepatic cells where it is re-phosphorylated by hexokinase. Since muscle
and brain cells lack glucose-6-phosphatase, the glucose-6-phosphate product of
hexokinase is retained and oxidized by these tissues.
In opposition to the cellular
responses to glucagon (and epinephrine on hepatocytes), insulin stimulates
extrahepatic uptake of glucose from the blood and inhibits glycogenolysis in
extrahepatic cells and conversely stimulates glycogen synthesis. As the glucose
enters hepatocytes it binds to and inhibits glycogen phosphorylase activity.
The binding of free glucose stimulates the de-phosphorylation of phosphorylase
thereby, inactivating it. Why is it that the glucose that enters hepatocytes is
not immediately phosphorylated and oxidized? Liver cells contain an isoform of
hexokinase called glucokinase. Glucokinase has a much lower affinity for
glucose than does hexokinase. Therefore, it is not fully active at the
physiological ranges of blood glucose. Additionally, glucokinase is not
inhibited by its product G6P, whereas, hexokinase is inhibited by G6P.
One major response of non-hepatic
tissues to insulin is the recruitment, to the cell surface, of glucose
transporter complexes. Glucose transporters comprise a family of five members, GLUT-1 to GLUT-5. GLUT-1 is
ubiquitously distributed in various tissues. GLUT-2 is found primarily in
intestine, kidney and liver. GLUT-3 is also found in the intestine and GLUT-
Hepatocytes, unlike most other cells,
are freely permeable to glucose and are, therefore, essentially unaffected by
the action of insulin at the level of increased glucose uptake. When blood
glucose levels are low the liver does not compete with other tissues for
glucose since the extrahepatic uptake of glucose is stimulated in response to
insulin. Conversely, when blood glucose levels are high extrahepatic needs are
satisfied and the liver takes up glucose for conversion into glycogen for
future needs. Under conditions of high blood glucose, liver glucose levels will
be high and the activity of glucokinase will be elevated. The G6P produced by
glucokinase is rapidly converted to G1P by phosphoglucomutase, where it can
then be incorporated into glycogen.
back to the
top
Digestion & Absorption of Proteins & Carbohydrates
Digestion and Absorption of Proteins
General Information:
1. Humans must ingest proteins, carbohydrates and
lipids to maintain tissue and organ functions.
2. Most of these nutrients consist of large
polymers that must be broken down before they can be made available to the
intestinal cells for transport.
3. Dietary proteins are cleaved by hydrolases
with specificity for the peptide bond (peptidases).
4. Endopeptidases (aka Proteases):
attack internal protein bonds liberating large peptide fragments.
Exopeptidases: cleave off one
amino acid at a time from the....
NH3+, aminopeptidases
or COO- terminus, carboxypeptidase.
5. Endo- and Exopeptidases work in concert
The Big Picture:
Protein Digestion and Absorption
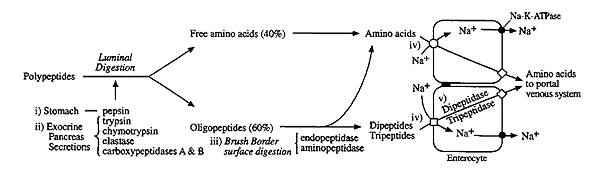
Gastric
(Stomach) Digestion:
1. Gastric HCl is responsible for the low
pH <2 of gastric juice.
2. Gastric acid kills microorganisms and denatures
dietary proteins preparing them for hydrolysis by proteases.
3. Gastric juices contain the acid stable
proteases of the pepsin family, which produce large peptide fragments
and some free amino acids.
4. Protein digestion at this stage is partial, as
the amino acids enter the duodenum, they trigger the release of
cholectystokinin-pancreozymin (CCK-PZ) into the bloodstream.
This release initiates the secretion of protease
zymogens from the pancreas and releases of enteropeptidase in
the gut.
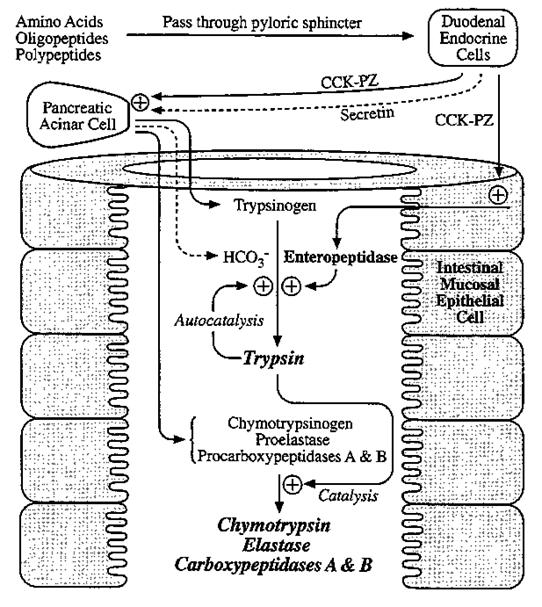
Pancreatic Proteases:
1. The pancreatic juice is rich in the proenzymes
of endopeptidase and carboxypeptidases.
2. Enteropeptidase activates
pancreatic trypsinogen to trypsin.
3. Trypsin autocatalytically activates
more trypsinogen and other proenzymes, liberating chymotrypsin,
elastase and the carboxypeptidases A and B.
Secretion and Activation of Pancreatic Proteases:
Digestion at the Brush Border
(surface of intestinal epithelial cells):
1. Since pancreatic juice does not contain
appreciable aminopeptidase activity, final digestion of di- and small peptides
depends on brush border enzymes.
2. The surface of intestinal epithelial cells is
rich in endopeptidases and aminopeptidases.
3. The end products of cell surface digestion are
free amino acids and di- and tripeptides.
Absorption:
1. Following digestion, amino acids and small
peptides are co-absorbed w/ sodium via group specific amino acid
or peptide transport systems.
2. These processes are carrier mediated,
discriminating between natural, L amino acids and D-amino acids, require
energy (from the Na+ gradient, Na-K ATPase) and physiologic
temperatures.
At least five brush border transport systems
exist:
1. neutral amino acids (uncharged aliphatic and
aromatic)
2. basic amino acids (
3. acidic amino acids (Asp, Glu)
4. imino acids (Pro), Hydroxyproline)
5. di- and tripeptides
Clinical Correlates:
1. Hartnup Disease:
Genetic defect in the neutral amino acid
transporter.
Symptoms: dermatitis due to tryptophan
malabsorption ("niacin" flush)
Consequences: not serious di- and tripeptide
absorption supply minimal amounts of dietarily essential neutral amino acids.
2. Cystinuria:
Precursor to kidney stones
Symptoms: painful kidney stone formation due to
malabsoprtion of cystine (two disulfide linked cysteines)
3. Sprue:
Destruction and flattening of the intestinal
villi resulting in generalized malabsorption.
Causes: bacterial infection or gluten (contained in
certain grains such as wheat and barley) sensitivity.
Digestion and Absorption of
Carbohydrates
General Information:
1. Carbohydrates provide a major component of the
daily caloric requirement, ~40%.
2.Distinguish between mono-, di- and
polysaccharides.
Monosaccharides- do not need
hydrolysis prior to absorption.
Disaccharides- require brush
border enzymes.
Polysaccharides- require brush
border enzymes, as well as, pancreatic amylase and salivary amylase for
digestion.
Starch:
Hydrolyzed by -amylase
into Maltotriose, -Limit
Dextrin, Maltose, Glucose
-1,4-glucosidic
linkages (non-branching, amylose) and branched chains -1,6 linkages (branch points,
amylopectin)
-amylase:
Present in saliva and pancreatic juice.
Specific for internal -1,4-glucosidic bonds.
Brush Border Carbohydrate Digestion:
Final hydrolysis of di- and oligosaccharides to monosaccharides is
carried out by -glucosidases
on the surface of the small intestine.
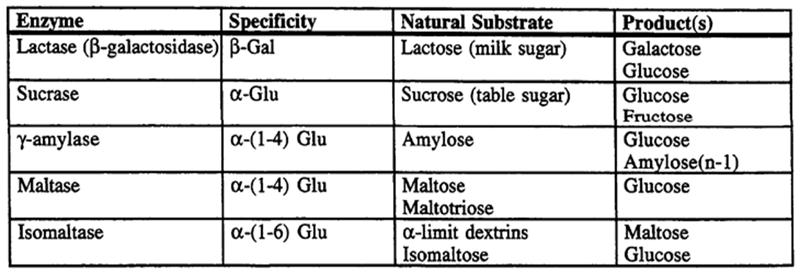
Monosaccharides are absorbed by carrier mediated transport.
At least two types are known:
1. Na+monosaccharide transporter
2. Na+ independent, diffusion type monosaccharide transport
system
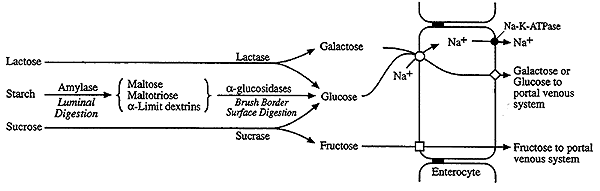
Undigested Carbohydrates:
1. Di-, oligo- and polysaccharides that are not hydrolyzed by -amylase and/or brush border enzymes
cannot be absorbed.
2. These carbohydrates reach the lower tract of the intestine
which contains bacteria.
3. The bacteria utilize many of the remaining carbohydrate, metabolizing
them and producing by- products such as: hydrogen gas, methane and carbon
dioxide.

Carbohydrates
Visualize Cell Pathways -
Turn research data into Pathways!
Free Software Trial - Download Now (www.stratagene.com) The field of glycomics -
Glycochemistry & Glycobiology
Read the Insight in Nature (www.nature.com/nature)
Mass Spectrometry Service -
Characterisation of Antibodies,
Glycoproteins & Proteins (GLP/GMP) (www.m-scan.com)
Carbohydrates
are one of three macronutrients that provide the body with energy (protein
and fats being the other two). The chemical compounds in carbohydrates are
found in both simple and complex forms, and in order for the body to use
carbohydrates for energy, food must undergo digestion, absorption, and glycolysis.
It is recommended that 55 to 60 percent of caloric intake come from
carbohydrates.
Chemical Structure
Carbohydrates
are a main source of energy for the body and are made of carbon, hydrogen, and oxygen.
Chlorophyll in plants absorbs light energy from the sun. This energy is used in
the process of photosynthesis, which allows green plants to take in carbon
dioxide and release oxygen and allows for the production of carbohydrates. This
process converts the sun's light energy into a form of chemical energy useful
to humans. Plants transform carbon dioxide (CO2) from the air, water
(H2O) from the ground, and energy from the sun into oxygen (O2)
and carbohydrates (C6H12O6) (6 CO2 +
6 H2O + energy = C6H12O6 + 6 O2).
Most carbohydrates have a ratio of 1:2:1 of carbon, hydrogen, and oxygen,
respectively.
Humans
and other animals obtain carbohydrates by eating foods that contain them. In
order to use the energy contained in the carbohydrates, humans must metabolize,
or break down, the structure of the molecule in a process that is opposite that
of photosynthesis. It starts with the carbohydrate and oxygen and produces
carbon dioxide, water, and energy. The body utilizes the energy and water and
rids itself of the carbon dioxide.
Simple Carbohydrates
Simple
carbohydrates, or simple sugars, are composed of monosaccharide or disaccharide
units. Common monosaccharides (carbohydrates composed of single sugar units)
include glucose, fructose, and galactose. Glucose is the most common
type of sugar and the primary form of sugar that is stored in the body for
energy. It sometimes is referred to as blood sugar or dextrose and is of
particular importance to individuals who have diabetes or hypoglycemia.
Fructose, the primary sugar found in fruits, also is found in honey and
high-fructose corn syrup (in soft drinks) and is a major source of sugar in the
diet of Americans. Galactose is less likely than glucose or fructose to
be found in nature. Instead, it often combines with glucose to form the
disaccharide lactose, often referred to as milk sugar. Both fructose and
galactose are metabolized to glucose for use by the body.
Oligosaccharides
are carbohydrates made of two to ten monosaccharides. Those composed of two
sugars are specifically referred to as disaccharides, or double sugars. They
contain two monosaccharides bound by either an alpha bond or a beta bond. Alpha
bonds are digestible by the human body, whereas beta bonds are more difficult
for the body to break down.
There
are three particularly important disaccharides: sucrose, maltose, and lactose.
Sucrose is formed when glucose and fructose are held together by an alpha bond.
It is found in sugar cane or sugar beets and is refined to make granulated
table sugar. Varying the
degree of purification alters the
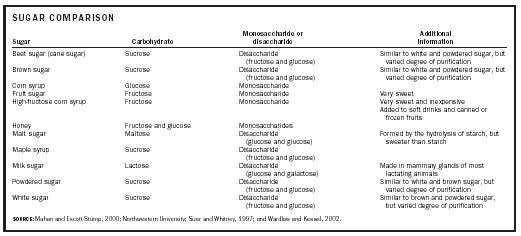
SUGAR
COMPARISON
|
Sugar |
Carbohydrate |
Monosaccharide
or disaccharide |
Additional
information |
|
Beet
sugar (cane sugar) |
Sucrose |
Disaccharide
(fructose and glucose) |
Similar to white and powdered sugar, but varied
degree of purification |
|
Brown
sugar |
Sucrose |
Disaccharide
(fructose and glucose) |
Similar to white and powdered sugar, but varied
degree of purification |
|
Corn
syrup |
Glucose |
Monosaccharide
|
|
|
Fruit
sugar |
Fructose |
Monosaccharide
|
Very
sweet |
|
High-fructose
corn syrup |
Fructose |
Monosaccharide
|
Very sweet and inexpensive |
|
Honey |
Fructose
and glucose |
Monosaccharides
|
|
|
Malt
sugar |
Maltose |
Disaccharide
(glucose and glucose) |
Formed by the hydrolysis of starch, but sweeter
than starch |
|
Maple
syrup |
Sucrose |
Disaccharide
(fructose and glucose) |
|
|
Milk
sugar |
Lactose |
Disaccharide
(glucose and galactose) |
Made in mammary glands of most lactating
animals |
|
Powdered
sugar |
Sucrose |
Disaccharide
(fructose and glucose) |
Similar to white and brown sugar, but varied
degree of purification |
|
White
sugar |
Sucrose |
Disaccharide
(fructose and glucose) |
Similar to brown and powdered sugar, but varied
degree of purification |
|
SOURCE: Mahan and Escott-Stump, 2000;
Northwestern University; Sizer and Whitney, 1997; and Wardlaw and Kessel,
2002. |
|||
final
product, but white, brown, and powdered sugars all are forms of sucrose.
Maltose, or malt sugar, is composed of two glucose units linked by an alpha
bond. It is produced from the chemical decomposition of starch, which occurs
during the germination of seeds and the production of alcohol. Lactose is a
combination of glucose and galactose. Because it contains a beta bond, it is
hard for some individuals to digest in large quantities. Effective digestion
requires sufficient amounts of the enzyme lactase.
Complex Carbohydrates
Complex carbohydrates, or polysaccharides, are
composed of simple sugar units in long chains called polymers. Three
polysaccharides are of particular importance in human nutrition: starch,
glycogen, and dietary fiber.
Starch and glycogen are digestible forms of complex
carbohydrates made of strands of glucose units linked by alpha bonds. Starch,
often contained in seeds, is the form in which plants store energy, and there
are two types: amylose and amylopectin. Starch represents the main type of
digestible complex carbohydrate. Humans use an enzyme to break down the bonds
linking glucose units, thereby releasing the sugar to be absorbed into the
bloodstream. At that point, the body can distribute glucose to areas that need
energy, or it can store the glucose in the form of glycogen.
Glycogen is the polysaccharide used to store energy in
animals, including humans. Like starch, glycogen is made up of chains of
glucose linked by alpha bonds; but glycogen chains are more highly branched
than starch. It is this highly branched structure that allows the bonds to be
more quickly broken down by enzymes in the body. The primary storage sites for
glycogen in the human body are the liver and the muscles.
Another type of complex carbohydrate is dietary fiber.
In general, dietary fiber is considered to be polysaccharides that have not been
digested at the point of entry into the large intestine. Fiber contains sugars
linked by bonds that cannot be broken down by human enzymes, and are therefore
![Pastas and whole-grain breads contain complex carbohydrates, which are long strands of glucose molecules. Nutritionists recommend that 55–60 percent of calories come from carbohydrates, and especially complex carbohydrates. [Photograph by James Noble. Corbis. Reproduced by permission.]](Introduction%20to%20%20metabolism.%20Investigation%20of%20aerobic%20and%20anaerobic%20oxidation%20of%20glucose.files/image032.jpg)
Pastas and whole-grain breads contain
complex carbohydrates, which are long strands of glucose molecules.
Nutritionists recommend that 55–60 percent of calories come from carbohydrates,
and especially complex carbohydrates.
[Photograph by James Noble. Corbis.
Reproduced by permission.]
labeled as indigestible. Because of
this, most fibers do not provide energy for the body. Fiber is derived from
plant sources and contains polysaccharides such as cellulose,
hemicellulose, pectin, gums, mucilages, and lignins.
The indigestible fibers cellulose, hemicellulose, and
lignin make up the structural part of plants and are classified as insoluble
fiber because they usually do not dissolve in water. Cellulose is a nonstarch
carbohydrate polymer made of a straight chain of glucose molecules
linked by beta bonds and can be found in whole-wheat flour, bran, and
vegetables. Hemicellulose is a nonstarch carbohydrate polymer made of glucose,
galactose, xylose, and other monosaccharides; it can be found in bran and whole
grains. Lignin, a noncarbohydrate polymer containing alcohols and acids, is a
woody fiber found in wheat bran and the seeds of fruits and vegetables.
In contrast, pectins, mucilages, and gums are
classified as soluble fibers because they dissolve or swell in water. They are
not broken down by human enzymes, but instead can be metabolized (or fermented)
by bacteria present in the large intestine. Pectin is a fiber made of
galacturonic acid and other monosaccharides. Because it absorbs water and forms
a gel, it is often used in jams and jellies. Sources of pectin include citrus
fruits, apples, strawberries, and carrots. Mucilages and gums are similar in
structure. Mucilages are dietary fibers that contain galactose, manose, and
other monosaccharides; and gums are dietary fibers that contain galactose,
glucuronic acid, and other monosaccharides. Sources of gums include oats, legumes,
guar, and barley.
Digestion and Absorption
Carbohydrates
must be digested and absorbed in order to transform them into energy that can
be used by the body. Food preparation often aids in the digestion process. When
starches are heated, they swell and become easier for the body to break down.
In the mouth, the enzyme amylase, which is contained in saliva, mixes with food
products and breaks some starches into smaller units. However, once the
carbohydrates reach the acidic environment of the stomach, the amylase is
inactivated. After the carbohydrates have passed through the stomach and into
the small intestine, key digestive enzymes are secreted from the pancreas and the
small intestine where most digestion and absorption occurs. Pancreatic amylase
breaks starch into disaccharides and small polysaccharides, and enzymes from
the cells of the small-intestinal wall break any remaining disaccharides into
their monosaccharide components. Dietary fiber is not digested by the small
intestine; instead, it passes to the colon unchanged.
Sugars
such as galactose, glucose, and fructose that are found naturally in foods or
are produced by the breakdown of polysaccharides enter into absorptive
intestinal cells. After absorption, they are transported to the liver where
galactose and fructose are converted to glucose and released into the
bloodstream. The glucose may be sent directly to organs that need energy, it
may be transformed into glycogen (in a process called glycogenesis) for storage
in the liver or muscles, or it may be converted to and stored as fat.
Glycolysis
http://www.youtube.com/watch?v=O5eMW4b29rg&feature=related
The
molecular bonds in food products do not yield high amounts of energy when
broken down. Therefore, the energy contained in food is released within cells
and stored in the form of adenosine triphosphate (ATP), a high-energy compound
created by cellular energy-production systems. Carbohydrates are metabolized
and used to produce ATP molecules through a process called glycolysis.
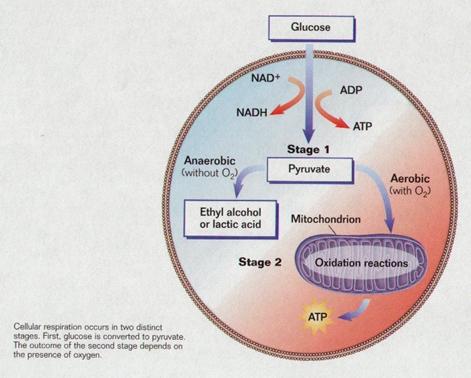
Glycolysis
breaks down glucose or glycogen into pyruvic acid through enzymatic
reactions within the cytoplasm of the cells. The process results in the
formation of three molecules of ATP (two, if the starting product was glucose).
Without the presence of oxygen, pyruvic acid is changed to lactic acid,
and the energy-production process ends. However, in the presence of oxygen,
larger amounts of ATP can be produced. In that situation, pyruvic acid is
transformed into a chemical compound called acetyle coenzyme A, a
compound that begins a complex series of reactions in the Krebs Cycle
and the electron transport system. The end result is a net gain of up to
thirty-nine molecules of ATP from one molecule of glycogen (thirty-eight
molecules of ATP if glucose was used). Thus, through certain systems, glucose
can be used very efficiently in the production of energy for the body.
Recommended Intake
At
times, carbohydrates have been incorrectly labeled as "fattening."
Evidence actually supports the consumption of more, rather than less, starchy
foods. Carbohydrates have four calories per gram, while dietary fats
contribute nine per gram, so diets high in complex carbohydrates are likely to
provide fewer calories than diets high in fat. Recommendations are for 55 to 60
percent of total calories to come from carbohydrates (approximately 275 to
Low-Carb Diets
Low-carbohydrate
diets, such as the Atkins and
—Paula
Kepos
It
is important to consume a minimum amount of carbohydrates to prevent ketosis,
a condition resulting from the breakdown of fat for energy in the absence of carbohydrates.
In this situation, products of fat breakdown, called ketone bodies, build up in
the blood and alter normal pH balance. This can be particularly harmful
to a fetus. To avoid ketosis, daily carbohydrate intake should include a
minimum of 50 to
Exchange System
The
exchange system is composed of lists that describe carbohydrate, fat, and
protein content, as well as caloric content, for designated portions of
specific foods. This system takes into account the presence of more than one
type of nutrient in any given food. Exchange lists are especially useful for
individuals who require careful diet planning, such as those who monitor intake
of calories or certain nutrients. It is particularly useful for diabetics, for
whom carbohydrate intake must be carefully controlled, and was originally
developed for planning diabetic diets.
Diabetes, Carbohydrate-Modified Diets, and
Carbohydrate Counting
Diabetes
is a condition that alters the way the body handles carbohydrates. In terms of
diet modifications, diabetics can control blood sugar levels by appropriately
managing the carbohydrates, proteins, and fats in their meals. The amount of
carbohydrates, not necessarily the source, is the primary issue. Blood glucose
levels after a meal can be related to the process of food preparation, the
amount of food eaten, fat intake, sugar absorption, and the combination of
foods in the meal or snack.
One
method of monitoring carbohydrate levels—carbohydrate counting—assigns a
certain number of carbohydrate grams or exchanges to specific foods.
Calculations are used to determine insulin need, resulting in better
control of blood glucose levels with a larger variety of foods. Overall,
diabetic diets can include moderate amounts of sugar, as long as they are
carefully monitored.
Glycolysis (detailed)
Nomenclature of carbohydrates - the naming of
carbohydrates - much more information than most of us need
1. Glucose - a monosaccharide - what you measure
in your blood.
The chemical formula for glucose is C6H12O6.
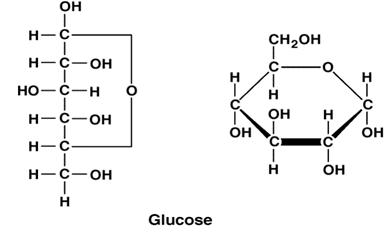
2. Sucrose - a disaccharide - table sugar. Is
made of one molecule of glucose and one fructose.

3. Starch - amylose and amylopectin - a polysaccharide
- potatoes.
Amylose is a straight chain of glucose molecules
linked together.
Amylopectin is a branched chain of glucose
molecules.
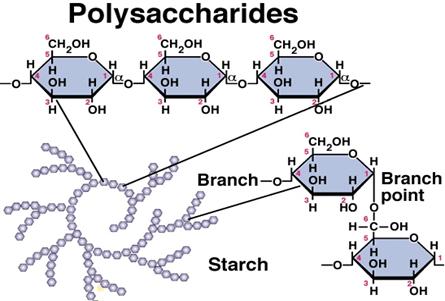
4. Glycolysis is the breakdown of glucose to
release its energy in the form of ATP.
Carbohydrate metabolism -
a slide presentation from the
more chemistry than most people will want.
From The Biology Home Page
produced by Jerry Johnson
from Frederick, Okl.